Verana Health Named RWE Leader After Strategic COTA Merger
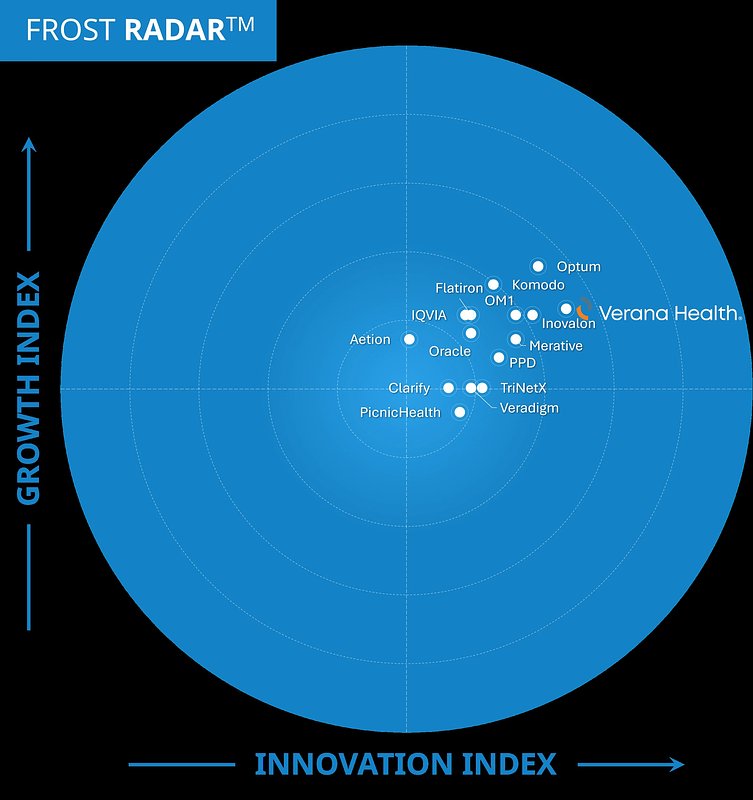
- 16 top-tier companies evaluated in the 2025 Frost Radar™ report, with Verana Health named the primary innovation leader.
- 17 of the top 20 global biopharmaceutical companies now served by Verana Health post-merger.
- 95 million de-identified patients included in Verana Health's expanded dataset, with over 10 million in oncology.
Experts agree that Verana Health's strategic merger with COTA and recognition as an RWE innovation leader solidify its role as a critical partner in accelerating drug development and improving patient care through high-quality, regulatory-grade data.
Verana Health's Rise: How a Data Powerhouse is Redefining Life Sciences
SAN FRANCISCO, CA – January 22, 2026 – By George Millen
In a significant validation of its strategic direction, digital health company Verana Health has been named the definitive innovation leader in the life sciences real-world evidence (RWE) market by Frost & Sullivan. The recognition, detailed in the prestigious 2025 Frost Radar™ report, places the company at the apex of a competitive field, citing its robust growth, technological prowess, and its delivery of high-fidelity, regulatory-grade data.
This accolade comes on the heels of a transformative merger with oncology data specialist COTA, a move that has dramatically expanded Verana Health's therapeutic footprint and solidified its position as an indispensable partner to the global biopharmaceutical industry. Together, these developments signal a major shift in the RWE landscape, with Verana Health emerging as a central force in accelerating drug development and improving patient care through the power of data.
"Verana Health's recognition on the Frost Radar reflects our commitment to innovation and service excellence, as we continue to significantly broaden and amplify our capabilities for the life sciences community," said Sujay Jadhav, CEO of Verana Health, in a statement. He credited the achievement to the company's use of artificial intelligence and advanced data curation to meet the complex needs of its partners.
A Crowded Field, A Clear Innovator
The Frost Radar™ for Life Sciences Real-World Evidence Solutions is a critical industry benchmark, evaluating market players on their ability to drive continuous innovation and translate it into consistent growth. For its 2025 report, Frost & Sullivan analyzed more than 45 vendors, ultimately shortlisting 16 top-tier companies, including established giants like IQVIA and specialized firms such as Flatiron Health and Aetion. To be named the primary innovation leader in such a crowded and dynamic market is a powerful endorsement.
Unmesh Lal, VP of Research at Frost & Sullivan and the report's lead author, highlighted the company's unique position. "Verana Health has rapidly positioned itself as an RWE innovation leader with a strong emphasis on delivering clinically rich, real-world data and insights, supported by trusted, research-ready evidence designed to meet the rigor required for clinical and regulatory decision-making," Lal stated.
The evaluation criteria for the Frost Radar are extensive, covering data quality, advanced analytics, scalability, and regulatory expertise. Verana Health's top ranking suggests it excels in turning vast, unstructured datasets into the high-quality evidence that life sciences companies depend on for everything from clinical trial design to post-market surveillance.
The Making of a Data Powerhouse
A key driver of Verana Health's ascent has been its aggressive and strategic expansion, most notably its recent merger with COTA. While Verana Health already held a commanding position in ophthalmology, urology, and neurology—bolstered by exclusive partnerships with the American Academy of Ophthalmology's IRIS® Registry and the American Urological Association's AQUA® Registry—the integration of COTA brought a deep and extensive oncology dataset into its portfolio.
This strategic fusion has created a data behemoth. The combined entity now serves 17 of the top 20 global biopharmaceutical companies, a testament to its critical role in the drug development ecosystem. Its data access has swelled to encompass insights from over 95 million de-identified patients, including more than 10 million in oncology, sourced from a network of over 20,000 clinicians and more than 30 Academic Medical Centers.
This scale is not just about size; it's about depth and breadth. By uniting these distinct, high-value specialty datasets, Verana Health can now offer a more holistic view of patient journeys and treatment outcomes across multiple critical therapeutic areas. This allows biopharma partners to address more complex research questions and accelerate development timelines in some of the most challenging areas of medicine.
The Gold Standard: Defining 'Regulatory-Grade' RWE
Central to Verana Health's value proposition is its focus on producing regulatory-grade RWE. This term signifies more than just a marketing buzzword; it refers to evidence derived from real-world data (RWD) that meets the stringent scientific and quality standards of regulatory bodies like the U.S. Food and Drug Administration (FDA). As regulators increasingly accept RWE to support drug approvals and label expansions, the demand for data that can withstand intense scrutiny has skyrocketed.
Achieving this standard requires a multi-faceted approach. It begins with high-quality data sources, such as the curated clinical registries Verana Health exclusively manages. These registries provide deep, longitudinal data directly from the point of care. However, raw data from electronic health records (EHRs) is often messy, incomplete, and unstructured. This is where Verana Health's technological innovation becomes critical.
Through advanced curation and AI-driven technologies, the company transforms this raw data into clean, structured, and research-ready datasets. This process involves harmonizing data from different sources, filling in gaps, and validating information to ensure its accuracy and reliability. The result is evidence that life sciences companies can use with confidence in their regulatory submissions, health economics research, and market-access strategies, knowing it is built on a foundation of quality and transparency.
Innovation at the Core: AI and Advanced Curation
At the heart of Verana Health's leadership position is its sophisticated technology stack. CEO Sujay Jadhav emphasized this, stating, "Through AI and advanced curation technology, we're delivering real-world insights across multiple specialty therapeutic areas to exceed our customers' needs and empower them to perform with confidence."
This technology acts as the engine that converts the massive potential of RWD into tangible, actionable intelligence. The company's AI algorithms are designed to read and interpret unstructured clinical notes, pathology reports, and other complex data types that are often locked away in EHR systems. By extracting and structuring this information, Verana Health unlocks a deeper level of clinical nuance—such as disease severity, treatment response, and specific patient characteristics—that is often missing from claims data or simpler data sources.
This AI-powered curation, guided by physician expertise, provides a significant competitive edge. It enables the company to produce the clinically rich data that Frost & Sullivan highlighted as a key strength. For pharmaceutical clients, this means access to more precise patient cohorts for clinical trial recruitment, a better understanding of treatment patterns in the real world, and the ability to generate evidence that more accurately reflects a drug's true value and impact. This fusion of unparalleled data access and cutting-edge technology is what solidifies Verana Health's role not just as a data provider, but as a true innovation partner in the quest for new therapies.
