Qlosi Eye Drops: A Precise Focus for Age-Related Blurry Vision
- 128 million Americans affected by presbyopia
- 0.4% pilocarpine concentration in Qlosi, compared to 1.25% in Vuity
- No serious adverse events reported with Qlosi in first 10 months of real-world use
Experts conclude that Qlosi's low-concentration, pupil-selective approach offers a safer, better-tolerated alternative to existing presbyopia treatments, with minimal ciliary muscle movement and a strong safety profile.
New Data Positions Qlosi as a Precise, Gentler Option for Blurry Near Vision
PONTE VEDRA, Fla. – January 27, 2026 – Millions of adults over 40 who struggle to read menus or phone screens may soon have a more refined treatment option. Orasis Pharmaceuticals has announced compelling new data for its presbyopia eye drop, Qlosi™, suggesting it offers a highly targeted approach to vision correction with a potentially improved safety and comfort profile compared to earlier treatments.
The findings, presented at the prestigious Hawaiian Eye & Retina 2026 Conference, reveal that Qlosi works with remarkable precision. A study from the world-renowned Bascom Palmer Eye Institute demonstrated that the low-concentration formula significantly improves near vision by constricting the pupil without engaging the eye’s focusing muscle—a key distinction that could set a new standard for patient comfort and safety in the rapidly growing market for presbyopia treatments.
The Science of a 'Pupil Selective' Approach
Presbyopia, the age-related loss of near-focusing ability, affects more than 128 million Americans. For years, the primary solutions were reading glasses or invasive surgery. The recent emergence of pharmaceutical eye drops has offered a new frontier of convenience. These drops typically use a class of drugs called miotics to shrink the pupil, creating a "pinhole effect" that increases the depth of focus, much like a camera aperture.
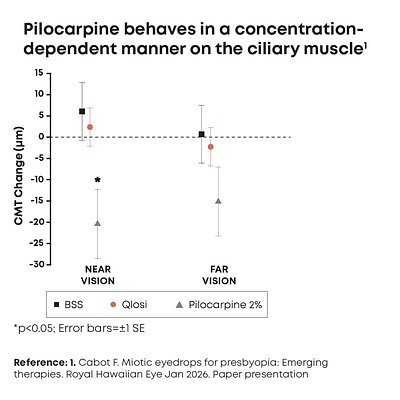
The active ingredient in Qlosi is pilocarpine, a long-established miotic. However, the new study highlights that not all pilocarpine formulations are created equal. The prospective, head-to-head clinical study used high-resolution optical coherence tomography (OCT) imaging to compare the effects of Qlosi (0.4% pilocarpine), a higher-concentration 2.0% pilocarpine solution, and a simple balanced salt solution (BSS) control.
The results were striking. While both pilocarpine concentrations effectively reduced pupil size, only the 2.0% solution caused a statistically significant contraction of the ciliary muscle—the muscle responsible for focusing. This muscle contraction is often linked to common side effects of miotic drops, such as headaches, brow ache, and blurred distance vision. In the study, Qlosi's effect on the ciliary muscle was "indistinguishable from the BSS control," according to Dr. Florence Cabot, Assistant Professor of Clinical Ophthalmology at Bascom Palmer Eye Institute.
"These findings are very encouraging, supporting that pilocarpine 0.4% is pupil selective and behaves in a concentration-dependent manner," Dr. Cabot stated in the announcement. She added that this reinforces how "low-concentration options may be particularly meaningful for patients considering a presbyopia therapy that balances both efficacy and safety."
Orasis CEO Elad Kedar emphasized that this data scientifically validates the real-world experience with the drug since its launch. "Qlosi demonstrating minimal ciliary muscle movement... is consistent with the results we have seen in real world experience to date—very low rates of adverse events overall without a single serious adverse event reported since launch," he said.
Navigating a New Market for Vision Correction
Qlosi enters a competitive landscape. AbbVie's Vuity, the first FDA-approved presbyopia drop, launched in 2021 and established the market. Vuity uses a 1.25% pilocarpine solution, more than three times the concentration of Qlosi. While it proved the viability of a pharmaceutical approach, some users reported side effects like headache and difficulty seeing in dim light.
Orasis's strategy appears to hinge on this differentiation. By developing its preservative-free EyeQ Formulation™ with the lowest effective concentration of pilocarpine, the company is positioning Qlosi as a refined, next-generation alternative. This "pupil selective" mechanism provides a powerful marketing message for both clinicians and patients who may be wary of potential side effects.
"The goal is not just to treat the condition, but to do so with the minimal necessary intervention," commented one independent optometrist who was not involved in the study. "If you can achieve the desired clinical effect on the pupil without causing off-target effects on the ciliary body, you're looking at a potentially cleaner, better-tolerated therapy. This is a significant evolution in the category."
The market is substantial and growing. With nearly two billion people affected globally, the demand for non-invasive, effective solutions is immense. The success of Qlosi will depend not only on its clinical profile but also on its market access, pricing, and ability to educate both doctors and patients on its unique benefits.
A Renewed Focus on Safety and Patient Comfort
The most critical differentiator for Qlosi may ultimately be its safety profile. While miotic drops are generally considered safe, rare but serious concerns, such as retinal detachment, have been reported with the use of miotics. This risk is theoretically linked to the forceful contraction of the ciliary muscle, which can create traction on the retina in susceptible individuals.
By demonstrating that Qlosi does not cause significant ciliary muscle movement, Orasis is directly addressing this paramount safety concern. The company reported that in the first 10 months of real-world use, there have been no reported cases of retinal detachment or other serious adverse events, according to data from the FDA Adverse Event Reporting System (FAERS).
This clean safety record, combined with a formulation that is preservative-free and includes dual lubricating agents, is designed to maximize patient comfort and long-term adherence. Preservatives in eye drops can cause irritation and ocular surface disease over time, making preservative-free options the preferred choice for many eye care professionals for chronic-use medications.
"Patient selection is still key," noted an ophthalmologist familiar with the drug class. "But having an option that minimizes ciliary side effects broadens the pool of appropriate candidates. It could be a game-changer for patients who were sensitive to other drops or hesitant to try them because of side-effect concerns."
As millions of people increasingly rely on screens for work and daily life, the frustrating blur of presbyopia is becoming a more prominent issue. The evolution of pharmaceutical treatments from a one-size-fits-all approach to more nuanced, targeted therapies like Qlosi represents a significant step forward. It offers the promise of restoring clear near vision not just effectively, but also gently, allowing individuals to shed their reading glasses without trading one inconvenience for another.
