New Era for Glaucoma Care? Adjustable Implant Begins US Trials
- 80 million people globally affected by glaucoma, a leading cause of irreversible blindness.
- Less than half of patients use glaucoma eye drops consistently after one year.
- 70 patients to be enrolled in the ADAPT trial to evaluate the Calibreye system's safety and effectiveness over 12 months.
Experts view the Calibreye™ Titratable Glaucoma Therapy™ as a potentially transformative advancement in glaucoma treatment, offering personalized, adjustable IOP control that could reduce complications and improve long-term outcomes for patients with refractory glaucoma.
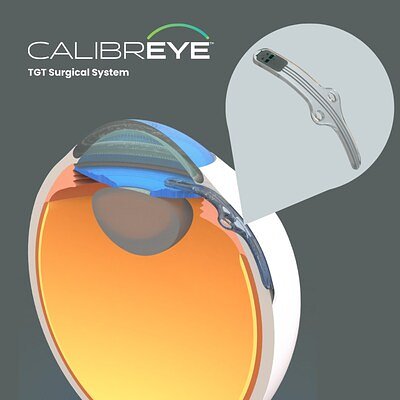
New Era for Glaucoma Care? Adjustable Implant Begins US Trials
CAMPBELL, CA – January 26, 2026 – A potentially transformative new chapter in the fight against glaucoma began today as Myra Vision announced the first patient has been treated in its U.S. clinical trial for the Calibreye™ Titratable Glaucoma Therapy™ (TGT) Surgical System. The milestone procedure, conducted by Dr. David Godfrey at Glaucoma Associates of Texas, marks a critical step forward for a device that promises to bring a new level of personalization to treating one of the world's leading causes of irreversible blindness.
The ADAPT clinical study will evaluate the safety and effectiveness of the Calibreye system, an innovative aqueous shunt designed to be adjustable after it has been surgically implanted. This event follows the U.S. Food and Drug Administration (FDA) granting an Investigational Device Exemption (IDE) in August 2025, paving the way for this crucial U.S. trial.
The Challenge of a Silent Thief
Glaucoma affects an estimated 80 million people globally, earning its reputation as the "silent thief of sight" by progressively and often painlessly damaging the optic nerve. With no cure available, treatment focuses on a single, critical goal: lowering intraocular pressure (IOP) to slow or halt the disease's progression.
For decades, the treatment pathway has been well-established, starting with eye drops and progressing to laser therapy or surgery. While medications can be effective, patient adherence is a persistent challenge, with studies showing that less than half of patients use their drops consistently after one year. When medication fails, physicians turn to surgical interventions.
Traditional procedures like trabeculectomy or the implantation of glaucoma drainage devices, also known as tube shunts, have been mainstays for managing moderate to severe glaucoma. More recently, a class of devices known as Minimally Invasive Glaucoma Surgery (MIGS) has offered a safer, less invasive alternative. However, nearly all of these surgical solutions share a common limitation: once the implant is in place, its drainage capacity is fixed. If the resulting IOP is too high or too low, the patient may face further complications or require additional surgeries. This static nature of treatment has long been a hurdle in the long-term management of a chronic, progressive disease.
A New Paradigm: Personalized IOP Control
Myra Vision's Calibreye system aims to shatter that static paradigm. Its defining feature is that it is "titratable," a term borrowed from chemistry that implies the ability to make small, precise adjustments. Once implanted, the Calibreye aqueous shunt is designed to allow clinicians to fine-tune fluid outflow from the eye during routine, postoperative follow-up appointments using a standard slit lamp.
This novel capability represents a fundamental shift from a one-size-fits-all approach to a dynamic, personalized one. It could empower doctors to adapt a patient's treatment as their individual clinical needs evolve over time, potentially avoiding the dangerous fluctuations in eye pressure that can accelerate vision loss.
"The potentially groundbreaking Calibreye System is the first adjustable glaucoma shunt in the United States, aiming to enable clinician-directed postoperative adjustment to support individualized intraocular pressure (IOP) management," said Dr. Godfrey, who performed the initial procedure. This in-office adjustability could significantly reduce the need for re-operations or a lifetime reliance on supplemental medications.
The Path to Approval: Inside the ADAPT Trial
The journey to bring such a novel device to patients is rigorous. The ADAPT trial is a prospective, multicenter, open-label study that will enroll up to 70 patients with refractory glaucoma—a form of the disease that has not responded to previous treatments. The study's primary goal is to evaluate the safety and effectiveness of the Calibreye system over a 12-month period.
"We are excited to have enrolled the first patient in our ADAPT clinical study. This is a significant accomplishment for our organization, and an important step towards delivering the Calibreye System to physicians and patients," commented Robert Chang, President and Chief Executive Officer of Myra Vision.
Chang noted that current filtration procedures are often reserved for a small fraction of eligible patients with advanced disease, partly due to the unpredictability and complication rates of older technologies. "The personalized IOP control provided by Calibreye will offer physicians greater confidence to address the needs of these underserved patients," he added.
The initiation of a U.S. clinical trial is the culmination of years of research and development, a point emphasized by one of the pioneers in the field.
"Having been involved in the development of this device for many years, it is incredibly rewarding to see our research come to fruition and now move into the U.S. clinical study," stated Ike Ahmed, M.D., a world-renowned glaucoma surgeon and a Myra Vision advisor. "Titratable glaucoma therapy has the potential to provide a meaningful solution for the significant number of these patients in need."
Navigating a Competitive MedTech Landscape
Myra Vision is a portfolio company of Shifamed, a highly specialized medical innovation hub known for identifying and accelerating the development of disruptive technologies. This backing provides Myra Vision with deep expertise in navigating the complex regulatory and commercial pathways required to bring a novel medical device to market.
The Calibreye system enters a competitive but rapidly evolving glaucoma surgical device market. It will be positioned against established traditional shunts and a growing array of MIGS devices from major players like Glaukos and Alcon. However, its unique adjustability could carve out a distinct and crucial niche, particularly for patients with moderate to advanced glaucoma who require significant and stable pressure reduction.
If the ADAPT trial successfully demonstrates that a titratable shunt is both safe and effective, it could not only secure FDA approval but also establish a new standard of care. The ability to customize treatment long after surgery is complete offers a new level of control in a disease defined by its relentless progression. For the millions living under the threat of irreversible vision loss, the results of the ADAPT study and the promise of personalized therapy represent a significant new beacon of hope.
