Neuvotion's AI Neurotech Aims to Rewire Stroke and SCI Recovery
- $1.5 billion: Global market value for neurorehabilitation devices in 2022, projected to exceed $4.8 billion by 2032. - $6 billion: Projected market size for brain-computer interface (BCI) technologies by 2030. - FDA 510(k) clearance (K240632): NeuStim™ received clearance in November 2024 for supporting hand movement recovery in stroke and spinal cord injury patients.
Experts view Neuvotion's AI-guided neurostimulation technology as a promising, non-invasive advancement in neurorehabilitation, with potential to enhance recovery outcomes through precision and personalized therapy.
Neuvotion's AI Neurotech Aims to Rewire Stroke and SCI Recovery
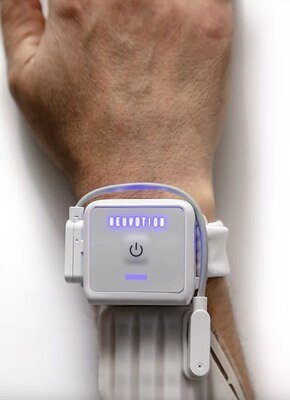
STAMFORD, Conn. – April 23, 2026 – Early-stage medical device company Neuvotion, Inc. today announced the upcoming launch of its AI-guided brain-body interface platform, NeuStim™, a system designed to revolutionize physical therapy for individuals recovering from stroke and spinal cord injuries. The FDA-cleared device offers a non-invasive method for restoring movement, positioning the company as a significant new entrant in the rapidly expanding neurotechnology market.
NeuStim™ provides high-resolution electrical stimulation to muscles, aiming to promote neuroplasticity—the brain's ability to reorganize itself by forming new neural connections. For the millions of people worldwide whose lives are profoundly altered by neurological damage, this technology represents a new frontier of hope for regaining critical functions, particularly in the hands.
"At Neuvotion we are extremely excited to help create new markets and applications for brain-computer interface and brain-body interface technologies," said Dr. Chad Bouton, Neuvotion's founder and CEO, in a statement. The company's platform launch marks a critical step in translating decades of advanced research into a tangible clinical tool.
A New Interface for Neurorecovery
At the core of Neuvotion's innovation is a user-centric design that addresses long-standing challenges in rehabilitation therapy. Traditional electrical stimulation often involves a cumbersome process of manually placing and replacing electrodes to find the optimal target on a muscle. NeuStim™ streamlines this with an innovative touchscreen interface that allows a clinician to electronically move the stimulation point across a grid of electrodes in real-time, pinpointing the most effective location within minutes.
This precision is key. The device received 510(k) clearance from the U.S. Food and Drug Administration (FDA) in November 2024 (K240632), specifically for supporting hand movement recovery in adult patients who have experienced a stroke resulting in hemiplegia (paralysis on one side of the body) or a spinal cord injury at the C-5 level. This clearance validates the device as safe and substantially equivalent to existing technologies, yet its feature set aims to surpass them in efficiency and efficacy.
Once a target is identified, clinicians can use a graphical user interface on a lightweight mobile device to set up and save personalized stimulation profiles for each patient. This combination of a surgery-free, wearable device with an intuitive, AI-guided setup promises to make advanced neurostimulation more accessible and efficient for therapy clinics.
Navigating a Booming Market
Neuvotion is entering a highly dynamic and competitive field. The global market for neurorehabilitation devices was valued at over $1.5 billion in 2022 and is projected to exceed $4.8 billion by 2032, driven by an aging population and a rising prevalence of neurological disorders. Within this space, brain-computer interface (BCI) and neurostimulation technologies are experiencing explosive growth, with the BCI market alone projected to reach over $6 billion by 2030.
The competitive landscape includes established medical device giants like Medtronic and Abbott, which have a strong foothold in implantable neurostimulators, as well as innovators in rehabilitation robotics like Ekso Bionics. However, Neuvotion’s NeuStim™ carves out a distinct niche. Unlike highly invasive BCIs from companies like Synchron or Neuralink, NeuStim™ is non-invasive, drastically lowering the barrier to entry for patients and providers. Compared to other non-invasive functional electrical stimulation (FES) systems, its AI-driven precision and streamlined workflow are significant differentiators.
One independent BCI researcher, speaking on the condition of anonymity, noted the strategic value of this approach. "The field is rightly excited about the potential of implanted brain interfaces, but the most immediate and widespread impact will likely come from non-invasive technologies that can be easily integrated into existing care models," the researcher explained. "A system that enhances what therapists can already do, while making it more precise and data-driven, is a very powerful proposition."
The Promise of Personalized Rehabilitation
Beyond the technology itself, the ultimate measure of NeuStim™'s success will be its impact on patient lives. Stroke and spinal cord injuries can be devastating, and the recovery process is often long and arduous, with progress plateauing over time. Neuvotion's platform is designed to make therapy more engaging and effective by tailoring stimulation to an individual's unique physiology and progress.
Experts in physical medicine believe AI-guided neuromodulation is the next logical step in rehabilitation. AI algorithms can analyze patient responses to stimulation and, over time, learn what patterns are most effective for promoting motor recovery. This creates a feedback loop that could potentially accelerate the process of rewiring the brain and restoring function more completely than traditional methods alone.
However, challenges remain. For any new medical technology, widespread adoption depends on robust clinical data demonstrating long-term efficacy, seamless integration into clinical workflows, and a viable cost structure for both clinics and patients. Neuvotion has already established a clinical research partnership with Good Shepherd Rehabilitation, a crucial step in gathering the real-world evidence needed to prove its value.
A Pipeline for the Future of Brain-Body Interface
NeuStim™ is just the first step in Neuvotion's broader vision. The company is already developing its next product, FocalStim™, which will target precise spinal cord stimulation to further enhance movement recovery. Furthermore, Neuvotion has signaled its ambition to create a full brain-body interface (BBI) system by also developing its own minimally-invasive BCI technology, bridging the gap between thought and action.
This ambitious roadmap is backed by a leadership team with deep expertise. Dr. Bouton's two decades of foundational research provide the scientific backbone, while the company's operational and regulatory efforts are led by industry veterans with extensive experience in commercializing medical devices. This blend of scientific vision and practical execution is critical for navigating the complex path from an early-stage concept to a market-leading solution.
As NeuStim™ prepares for its launch later this year, it represents more than just a new device; it embodies a pivotal shift toward more intelligent, personalized, and accessible tools for neurological recovery. By combining artificial intelligence with targeted neurostimulation, Neuvotion is aiming to empower both clinicians and patients, potentially rewriting the script for what is possible after a life-altering injury.
📝 This article is still being updated
Are you a relevant expert who could contribute your opinion or insights to this article? We'd love to hear from you. We will give you full credit for your contribution.
Contribute Your Expertise →