Hope for Becker MD: Sevasemten Halts Disease Decline in Long-Term Study
- NSAA Stability: Sevasemten-treated patients showed a minimal change of +0.1 on the NSAA scale over 3.5 years, compared to a predicted decline of -5.3 points. * Biomarker Reduction: Significant reductions in creatine kinase (CK), a marker of muscle damage, were observed. * Patient Enrollment: 99% of eligible participants in prior trials chose to enroll in the MESA extension study.
Experts view sevasemten as a groundbreaking therapy that could fundamentally alter the treatment landscape for Becker muscular dystrophy by stabilizing disease progression, marking the first potential approved treatment for this condition.
Hope for Becker MD: Sevasemten Halts Disease Decline in Long-Term Study
BOULDER, CO – March 10, 2026 – In a landmark development for the rare disease community, Edgewise Therapeutics (Nasdaq: EWTX) has presented long-term data suggesting its investigational drug, sevasemten, can halt the functional decline associated with Becker muscular dystrophy (BMD). The results, unveiled at the 2026 MDA Clinical and Scientific Conference, show that patients taking the oral therapy maintained their muscle function for up to 3.5 years, a stark contrast to the relentless progression typical of the genetic disorder, which currently has no approved treatments.
For decades, a diagnosis of Becker muscular dystrophy has meant an inevitable, albeit variable, decline in physical ability. The new data from the MESA open-label extension study offers the first significant hope that this trajectory can be altered. Participants treated with sevasemten demonstrated a remarkable stabilization of motor function, a finding that could herald a new era for thousands of patients globally.
"For the first time with an investigational agent, we are seeing long-term data where individuals with Becker are stable in the face of a disease that we have seen would otherwise lead to a significant functional decline," said Joanne Donovan, M.D., Ph.D., Chief Medical Officer of Edgewise Therapeutics, in a statement accompanying the release.
A New Horizon in a Devastating Disease
Becker muscular dystrophy is a genetic, X-linked neuromuscular disorder characterized by progressive muscle weakness and wasting. Caused by mutations in the dystrophin gene, the disease leads to contraction-induced muscle damage, irreversible muscle loss, and often, life-threatening cardiac complications. While typically less severe and slower to progress than Duchenne muscular dystrophy (DMD), its course is one of steady deterioration.
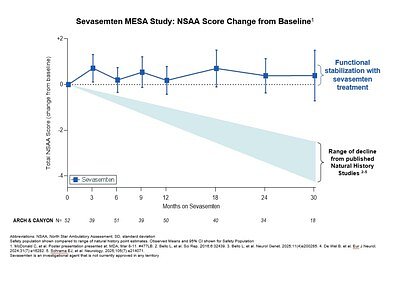
The clinical impact of the MESA study data is best understood through the North Star Ambulatory Assessment (NSAA), a 17-item scale that measures functional motor abilities. Natural history studies—which track disease progression without intervention—predict that Becker patients lose an average of 1.0 to 1.7 NSAA points per year. Over several years, this translates into a significant loss of independence and quality of life.
The sevasemten data paints a dramatically different picture. Key findings include:
- ARCH Study Cohort: Over 3.5 years, participants treated with sevasemten showed a minimal change of +0.1 on the NSAA scale. This stands in stark contrast to the predicted decline of -5.3 points for this same period based on natural history data.
- CANYON Study Cohort: Over 2 years, treated participants again showed a +0.1 improvement, versus a predicted decline of -2.9 points.
This divergence from the expected disease course is the most significant aspect of the findings, suggesting sevasemten is not merely slowing the decline but potentially stabilizing the disease in a sustained manner. Furthermore, the drug continued to demonstrate a favorable safety profile, a critical factor for any therapy intended for long-term use.
The Science of Stability
Unlike gene therapies or exon-skipping drugs that aim to restore the missing dystrophin protein, sevasemten employs a novel mechanism. It is a first-in-class, orally administered fast skeletal myosin inhibitor. In simple terms, it is designed to protect muscle fibers from the excessive damage caused by contractions when functional dystrophin is absent or insufficient.
By selectively modulating muscle contraction, the therapy aims to reduce the constant cycle of injury and inflammation that drives the disease's progression. The MESA study provides the strongest evidence yet that this approach is effective over the long term. The study itself is an open-label extension for patients who completed previous trials, and the fact that 99% of eligible participants chose to enroll speaks volumes about the perceived benefit and tolerability of the treatment among patients and their physicians.
Beyond functional stability, the data presented also showed significant reductions in key biomarkers of muscle damage, such as creatine kinase (CK). These biochemical indicators provide objective evidence that sevasemten is having its intended biological effect at the cellular level, reinforcing the functional outcomes observed in patients.
Navigating the Path to Approval
While the MESA data is exceptionally promising, the ultimate fate of sevasemten rests on the outcome of its pivotal, placebo-controlled trial, GRAND CANYON. Edgewise Therapeutics confirmed it is on track to announce top-line results from this crucial study in the fourth quarter of 2026. A positive outcome would be the final piece of the puzzle needed to support a marketing application to regulatory bodies like the U.S. Food and Drug Administration (FDA).
Edgewise has been strategic in its regulatory approach, securing multiple designations that could smooth and expedite the drug's path to market. Sevasemten has received Orphan Drug Designation from both the FDA and the European Medicines Agency (EMA), a status that provides incentives for developing treatments for rare conditions. It also holds FDA Fast Track designation, designed to facilitate the development and hasten the review of drugs that treat serious conditions and fill an unmet medical need.
Should GRAND CANYON succeed, sevasemten would be positioned to become the first-ever targeted therapy approved for Becker muscular dystrophy, fundamentally transforming the treatment landscape and setting a new standard of care.
Market Potential and Competitive Landscape
The financial markets have taken notice of sevasemten's potential. Edgewise Therapeutics' stock (EWTX) has seen significant positive momentum, with shares trading near 52-week highs. Analysts have responded favorably to the new data, with firms like Raymond James and Evercore ISI reiterating strong buy ratings and setting price targets that suggest substantial upside. This investor confidence is built on the vast unmet need in the Becker market and sevasemten's unique position as a potential first-in-class, first-to-market therapy.
While the broader muscular dystrophy space includes major players like Sarepta Therapeutics and PTC Therapeutics focused primarily on Duchenne, sevasemten's distinct mechanism and focus on Becker give it a clear and defensible market opportunity. Its approach of muscle protection could also position it as a foundational therapy, potentially for use in combination with other future treatments.
With a strong cash position of over $530 million reported at the end of 2025, Edgewise appears well-funded to see its lead candidate through the final stages of clinical development and a potential commercial launch. The entire Becker community, alongside investors and clinicians, now looks toward late 2026, when the results of the GRAND CANYON study could finally pave the way for a new era in the treatment of this relentless disease.
