FDA Fast-Tracks Novel Imaging Agent for Lung Disease Diagnosis
- 55% of ILD patients are misdiagnosed at least once before the true cause is identified.
- Fast Track Designation granted by the FDA to expedite development of 99mTc-maraciclatide.
- Phase II PREDICT-ILD trial preliminary data suggests feasibility of visualizing inflammation in fibrotic-ILD.
Experts agree that 99mTc-maraciclatide could revolutionize ILD diagnosis by providing a non-invasive method to distinguish inflammation from fibrosis, potentially improving treatment outcomes and reducing diagnostic delays.
FDA Fast-Tracks Novel Imaging Agent for Lung Disease Diagnosis
LONDON, UK – February 12, 2026 – The U.S. Food and Drug Administration (FDA) has granted Fast Track Designation to an investigational imaging agent that could revolutionize the diagnosis of a vast and life-threatening category of lung conditions. London-based Serac Healthcare announced today that its molecular imaging agent, 99mTc-maraciclatide, has been placed on an expedited regulatory pathway for its potential to visualize inflammation in patients with interstitial lung disease (ILD).
The designation marks a significant milestone for a diagnostic tool aimed at solving one of the most difficult challenges in pulmonology: seeing the difference between active, treatable inflammation and permanent, irreversible scarring within the lungs.
The Agonizing Path to an ILD Diagnosis
Interstitial lung disease is not a single ailment but a complex umbrella term for over 200 distinct disorders characterized by progressive lung damage. For patients, the journey to a correct diagnosis is often long and fraught with uncertainty. Initial symptoms like a persistent dry cough and shortness of breath are non-specific and easily mistaken for more common conditions like asthma or heart failure. Research shows that this ambiguity leads to significant diagnostic delays, with many patients waiting a year or more and up to 55% being misdiagnosed at least once before the true cause is identified.
Currently, the cornerstone of ILD diagnosis is a high-resolution computed tomography (HRCT) scan of the chest, which provides detailed anatomical images. While HRCT can identify patterns of fibrosis, or scarring, it struggles to definitively distinguish it from active inflammation. This distinction is clinically vital. Inflammatory ILDs may respond well to powerful anti-inflammatory or immunosuppressive therapies. However, administering these same drugs to a patient whose disease is predominantly fibrotic, like in Idiopathic Pulmonary Fibrosis (IPF), can be ineffective and even harmful.
To gain certainty, doctors may turn to a surgical lung biopsy, an invasive procedure that carries significant risks, particularly for patients with already compromised lung function. The medical community has long recognized the profound unmet need for a non-invasive tool that can accurately differentiate inflammation from fibrosis, enabling doctors to make earlier, more confident treatment decisions.
A New Way to See Inside the Lungs
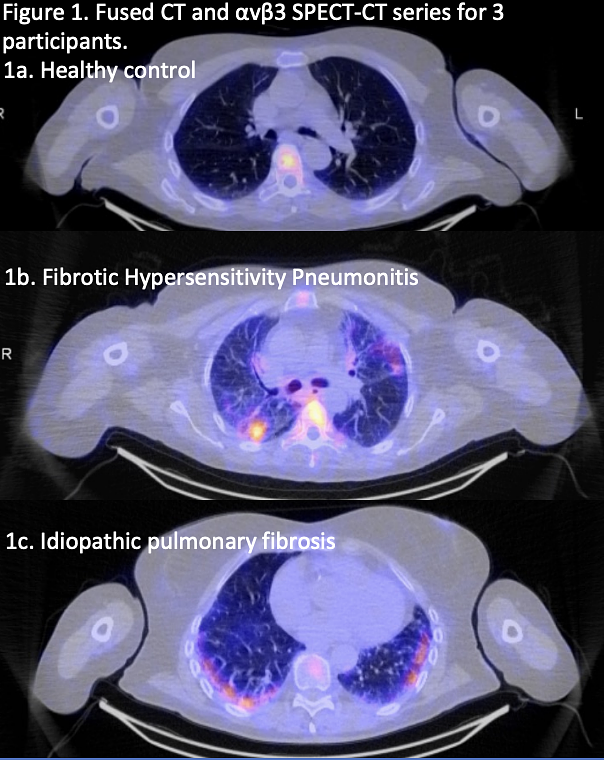
Serac Healthcare's 99mTc-maraciclatide aims to be that tool. It represents a leap forward in molecular imaging, a field of medicine that uses targeted agents to visualize biological processes at the cellular level. The agent is a radio-labelled tracer designed to perform a highly specific task: find and bind to a molecule called αvβ3 integrin.
This integrin is a type of cell-adhesion molecule that is not abundant in healthy, stable tissue. However, its expression skyrockets on the surface of endothelial cells during angiogenesis—the formation of new blood vessels—which is a key biological process that fuels inflammation. By tagging a molecule that binds to αvβ3 integrin with a safe, short-lived radioactive isotope (99m-Technetium), the agent effectively becomes a beacon for inflammation.
When injected into a patient, 99mTc-maraciclatide circulates through the body and accumulates in areas of the lung where inflammation is active. A subsequent SPECT-CT scan, which combines functional nuclear imaging with anatomical CT imaging, can then generate a detailed 3D map pinpointing the precise location and extent of this inflammatory activity. Preliminary data from the company's Phase II PREDICT-ILD trial have already demonstrated that visualizing inflammation in people with fibrotic-ILD could be feasible with this method. Full results from the trial are expected to be published later this year.
Navigating the Regulatory Fast Lane
The FDA's decision to grant Fast Track Designation acknowledges both the seriousness of ILD and the potential for 99mTc-maraciclatide to address a critical unmet medical need. This regulatory mechanism is designed to facilitate the development and expedite the review of drugs and diagnostics that show promise in treating or diagnosing serious conditions.
The benefits are substantial and are intended to reduce the time to potential approval and get the diagnostic into the hands of clinicians and patients more quickly. Under Fast Track, Serac Healthcare will have more frequent meetings and written communication with the FDA to discuss the agent's development plan and clinical trial design. It also enables a "Rolling Review," where the company can submit completed sections of its New Drug Application (NDA) for review by the FDA as they are completed, rather than waiting to submit the entire application at once. Furthermore, the designation opens the door to eligibility for Accelerated Approval and Priority Review down the line, should relevant criteria be met.
"The FDA's Fast Track designation of maraciclatide signals the imperative for improved ILD diagnosis, assessment, and monitoring," said David Hail, Chief Executive Officer of Serac Healthcare, in a prepared statement. He emphasized the difficulty of early detection due to non-specific symptoms and the potential for inappropriate treatment to be more detrimental than beneficial.
Potential to Reshape ILD Care
Should 99mTc-maraciclatide successfully navigate its clinical trials and gain regulatory approval, its impact could be transformative. A non-invasive, reliable method for distinguishing inflammation from fibrosis would empower clinicians to move beyond the current limitations of imaging and invasive biopsies. It could enable earlier and more precise diagnoses, leading to the selection of the right therapy for the right patient at the right time.
As David Hail noted, "A non-invasive imaging solution capable of distinguishing inflammation and fibrosis predominant ILD has the potential to meaningfully advance early diagnosis, change the treatment paradigm and improve patient outcomes." This could mean starting patients on targeted anti-fibrotic or anti-inflammatory medications sooner, monitoring their response to therapy with greater accuracy, and ultimately personalizing the management of this complex group of diseases.
This development arrives amidst a broader push within respiratory medicine towards precision diagnostics, with other research efforts focused on blood-based biomarkers and artificial intelligence algorithms for interpreting scans. The advancement of 99mTc-maraciclatide, however, represents a direct, functional approach to imaging a core pathological process. For the hundreds of thousands of people worldwide living with the uncertainty of an ILD diagnosis, this accelerated path forward offers a tangible sense of hope.
