Agitated Solutions Earns EU Approval for Stroke-Detecting Heart Device
- 25% of adults have right-to-left cardiac shunts, a leading cause of stroke.
- The European medical technology market is valued at over €170 billion.
- The Orbis™ device automates a 60-year-old manual process, improving consistency and diagnostic confidence.
Experts agree that the Orbis™ Microbubble Generator represents a significant advancement in cardiac diagnostics, offering improved consistency and efficiency in detecting cardiac shunts linked to stroke.
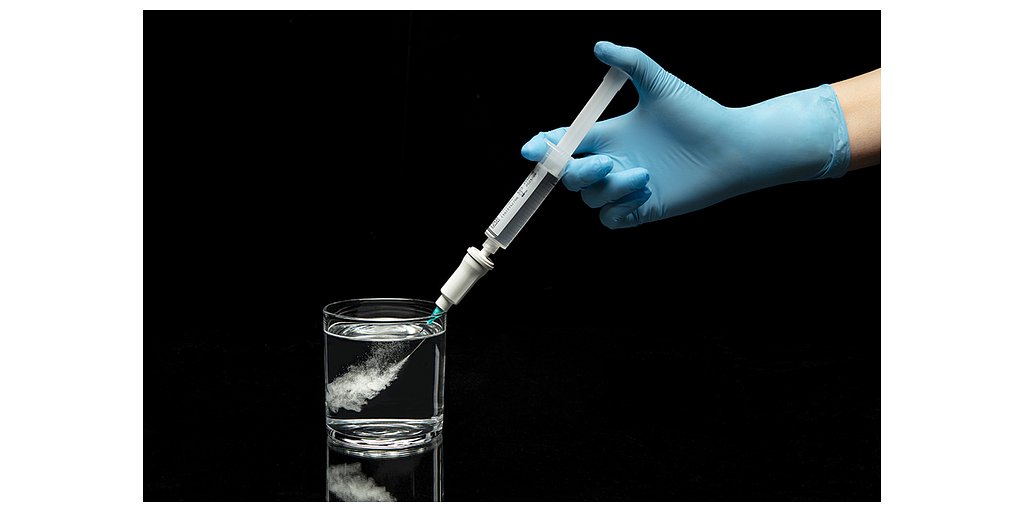
New Device to Modernize Cardiac Diagnostics Receives European Approval
ST. PAUL, Minn. – April 29, 2026 – Medical technology firm Agitated Solutions, Inc. has secured a pivotal CE Mark approval in the European Union for its Orbis™ Microbubble Generator. The certification clears the path for the novel device to enter the vast European healthcare market, offering a significant technological leap in the diagnosis of cardiac conditions linked to stroke.
The Orbis™ generator is designed to automate and standardize a common procedure known as a saline contrast echocardiography, or “bubble study.” For decades, clinicians have relied on this test to detect right-to-left cardiac shunts—small holes or passages in the heart, like a patent foramen ovale (PFO), that allow blood to flow improperly. These shunts are surprisingly common, affecting approximately one-quarter of the adult population, and are a leading cause of stroke, particularly in younger individuals.
“The CE Mark approval underscores the strength of our innovation and its potential to redefine the standard for saline microbubble contrast,” said Morgan Evans, Co-Founder and CEO of Agitated Solutions, in a statement. “We are excited to bring this solution to European clinicians, helping to improve consistency, streamlining procedures, and enhancing diagnostic confidence.”
A 60-Year-Old Problem Gets a Modern Solution
For nearly 60 years, the bubble study has been a cornerstone of cardiac diagnostics, but the process has remained stubbornly manual and variable. The procedure involves two clinicians: one vigorously agitates saline in a syringe to create microbubbles and injects them into the patient, while another operates an ultrasound machine to capture images of the bubbles traveling through the heart. The appearance of bubbles on the left side of the heart indicates a shunt.
The manual agitation process, however, is fraught with inconsistency. The size, concentration, and stability of the microbubbles can vary dramatically between tests and even between syringes in the same test. This variability can lead to suboptimal ultrasound images, making it difficult for cardiologists to make a definitive diagnosis.
“The lack of consistency impacts the ability to accurately diagnose and treat patients,” stated Dr. Micah Eimer, Chief Medical Officer of Agitated Solutions. “The Orbis™ Microbubble Generator is a major advancement towards providing clinicians a more consistent contrast and image quality, and facilitating downstream care.”
The Orbis™ device replaces this manual method with a controlled, automated system. It produces microbubbles of a consistent size and concentration, designed to generate clear, bright, and long-lasting ultrasound images. Critically, the system is designed to be operated by a single sonographer, freeing up a second clinician and addressing the resource-intensive nature of the traditional method. This efficiency gain is expected to streamline workflow in busy echocardiography labs.
Unlocking the European Market Amidst Regulatory Hurdles
Securing the CE Mark is a significant commercial milestone, opening the door to the European medical technology market, estimated at over €170 billion. The cardiovascular diagnostic and monitoring segment in Europe is particularly robust, with projections showing a compound annual growth rate of over 6%.
This achievement is especially noteworthy given the European Union's stringent Medical Device Regulation (MDR), which came into full effect in 2021. The MDR introduced a more rigorous and complex approval process, creating significant hurdles for manufacturers. Successfully navigating this framework serves as a strong validation of the Orbis™ device's safety, quality, and clinical documentation. For a company like Agitated Solutions, it signals to the market that its technology meets the highest contemporary standards.
A Global Vision Supported by Strategic Partnerships
The European approval is a key part of a broader global strategy for the Minnesota-based company. The Orbis™ Microbubble Generator is already approved and available in Canada, and the company is actively pursuing entry into the U.S. market. An Investigational New Drug (IND) study, known as ENHANCE (NCT06938347), is currently underway in the United States and Canada to support a submission to the U.S. Food and Drug Administration (FDA).
This trial is focused on the company's proprietary ultrasound contrast agent, ASI-02, which has received Fast Track designation from the FDA—a program designed to expedite the review of drugs and biologics that treat serious conditions and fill an unmet medical need. Agitated Solutions is developing a comprehensive platform that extends beyond the generator, including workflow accessories and AI-enabled software solutions to further enhance diagnostic capabilities.
This ambitious vision has attracted significant financial and strategic backing. The company recently completed a Series B financing round, notably joined by the venture arm of W. L. Gore & Associates. This partnership is highly strategic, as Gore is a global leader in medical devices, including those used to close the very PFOs that the Orbis™ system is designed to help diagnose. This alignment between a diagnostic innovator and a therapeutic leader underscores the industry's confidence in the clinical need and commercial potential of improving cardiac shunt detection.
📝 This article is still being updated
Are you a relevant expert who could contribute your opinion or insights to this article? We'd love to hear from you. We will give you full credit for your contribution.
Contribute Your Expertise →