NY Approves AI Psoriasis Test, Unlocking Nationwide Precision Care
- 91% positive predictive value: The Mind.Px test accurately predicts the most effective biologic drug class for psoriasis patients.
- $5,138 to $13,141 annual savings per patient: Potential cost reduction by avoiding trial-and-error biologic treatments.
- $63M raised: Mindera Health's total funding to date, supporting nationwide rollout.
Experts view the NY approval of Mind.Px as a major validation of AI-driven precision medicine in dermatology, offering a scientifically rigorous solution to optimize psoriasis treatment and reduce healthcare costs.
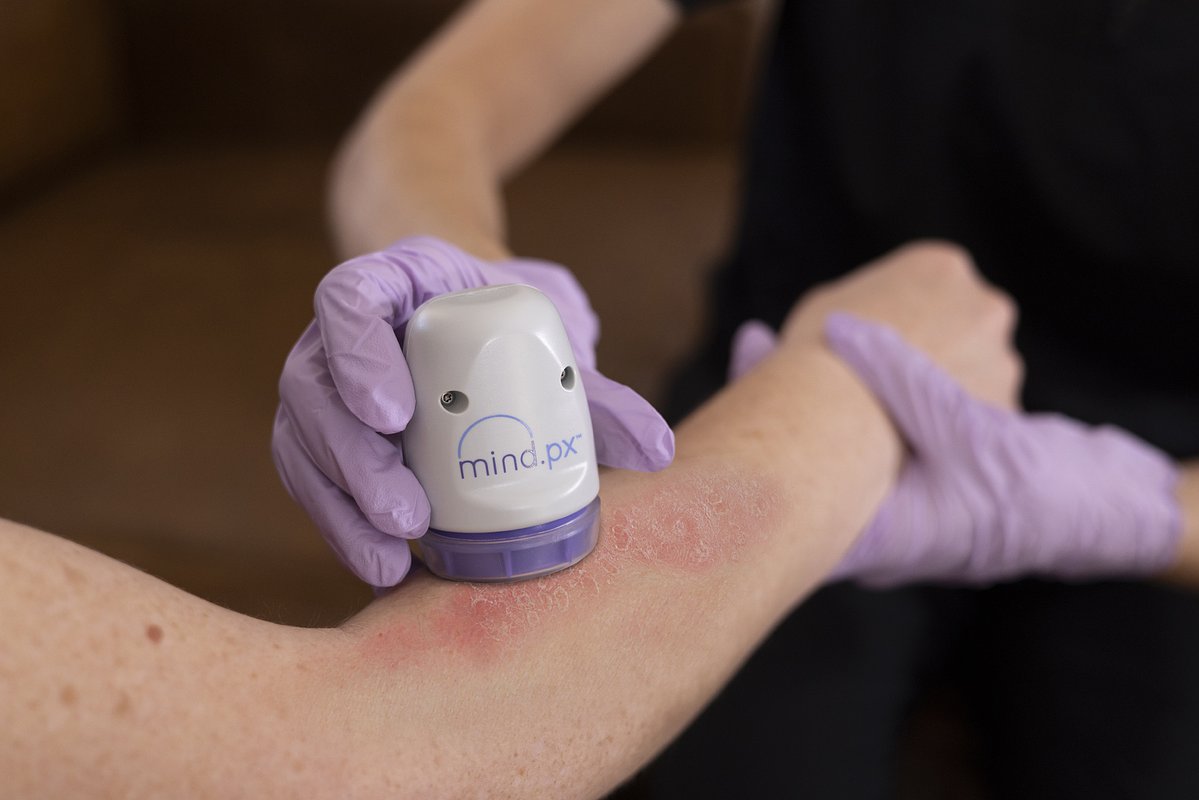
AI Psoriasis Test Gets NY Nod, Unlocking Nationwide Precision Care
VISTA, CA – April 22, 2026 – Mindera Health announced today it has secured approval from the New York State Department of Health for Mind.Px, a groundbreaking test designed to guide treatment for psoriasis. The approval is a critical milestone, signifying that the test has met one of the nation's most stringent regulatory benchmarks and is now available to patients in all 50 states.
A New Era for Psoriasis Treatment
Psoriasis, a chronic autoimmune condition affecting millions, has long been treated with a frustrating and costly "trial-and-error" approach. Patients with moderate-to-severe disease are often prescribed powerful biologic drugs, but with a 30-40% chance the first-choice medication will fail, leading to months of continued suffering and wasted expense.
Mindera Health aims to replace this guesswork with data-driven precision. The Mind.Px test utilizes a proprietary dermal biomarker patch that painlessly and non-invasively collects mRNA from a patient's skin. This genetic material is then analyzed using Next-Generation Sequencing (NGS), and the data is fed into a machine learning algorithm. The result is a prediction, with over 91% positive predictive value, of which class of biologic drug—such as TNFαi, IL-17i, or IL-23i—is most likely to be effective for that specific individual.
By providing this molecular-level insight before treatment begins, the test empowers clinicians to select a more targeted therapy from the start, dramatically increasing the odds of a successful outcome on the first try.
The Gold Standard of Approval
Securing approval from the New York State Department of Health's Clinical Laboratory Evaluation Program (CLEP) is a significant validation of Mindera Health's technology. CLEP is widely regarded in the medical industry as the gold standard for laboratory testing, with requirements that often exceed federal CLIA regulations. Any laboratory testing specimens from New York residents must pass this rigorous review, which scrutinizes everything from scientific validity and personnel qualifications to on-site processes.
"We are extremely proud to have met the quality standards established by New York’s Clinical Laboratory Evaluation Program (CLEP), a standard broadly seen as the highest quality benchmark for clinical laboratory and laboratory developed tests,” said Tobin Dickerson, Chief Scientific Officer and Co-Founder of Mindera Health. “This certification demonstrates the scientific rigor of our laboratory team and our flagship test, Mind.Px, and we are eager to bring precision medicine to the psoriasis patients of New York to help guide health care providers’ biologic selection.”
This approval serves as a powerful endorsement of the test's accuracy and reliability, a key factor for adoption by both clinicians and healthcare payers.
Tackling the Billion-Dollar Guessing Game
The financial implications of optimizing psoriasis treatment are staggering. The global market for psoriasis therapeutics is valued in the tens of billions, with projections to exceed $70 billion by the mid-2030s. Biologic drugs represent the largest and most expensive segment of this market, with annual costs for a single patient ranging from approximately $12,000 to over $80,000.
When the first-line biologic fails, the costs multiply. Patients endure prolonged symptoms while payers cover the expense of an ineffective drug, followed by the cost of a second, and sometimes third, option. A budget impact study published in SKIN The Journal of Cutaneous Medicine quantified the potential savings from using Mind.Px to guide this initial choice. The model predicted annual cost savings ranging from $5,138 to $13,141 per patient.
By reducing the need for sequential, trial-and-error prescriptions, the test presents a compelling value proposition for the entire healthcare ecosystem. It aligns the interests of patients, who achieve relief faster; physicians, who can prescribe with greater confidence; and insurance providers, who can curb wasteful spending on ineffective treatments. This economic benefit is expected to be a major driver in securing broader insurance coverage for the diagnostic.
From Lab Bench to Nationwide Access
The journey of Mind.Px from a concept to a nationally available diagnostic has been fueled by strategic investment and a clear vision for the future of dermatology. Mindera Health, a private company based in the San Diego area, has successfully raised over $63 million in funding to date. This includes a recent $14 million convertible note financing round in July 2024, signaling strong investor confidence in its commercialization strategy.
These funds have been instrumental in building out the company's CLIA-certified and CAP-accredited laboratory and scaling production of its proprietary dermal patch. While the immediate focus is on the U.S. rollout of Mind.Px for psoriasis, Mindera Health has a broader ambition. The company is actively developing its Mindera Dermal Intelligence Platform to create similar predictive tests for other chronic and inflammatory skin conditions, including eczema and psoriatic arthritis.
With the final regulatory hurdle for nationwide access now cleared, Mindera Health is positioned to make precision medicine a new standard of care in dermatology. For the millions of Americans living with the daily challenges of psoriasis, this shift from guesswork to a genetically informed treatment plan offers a new level of hope for faster, more effective, and lasting relief.
