A Year of Freedom: New Diabetes Tech Duo Promises Less Interruption
- 1-year CGM sensor: Eversense® 365 provides continuous glucose monitoring for a full year, eliminating frequent sensor changes.
- 85%+ time-in-range: Early adopters report achieving exceptional glucose control within their first week.
- 70% insurance coverage: 70% of commercial providers already cover the twiist system.
Experts view this integrated system as a significant advancement in diabetes care, offering unparalleled convenience and long-term stability, though they note a learning curve for some users.
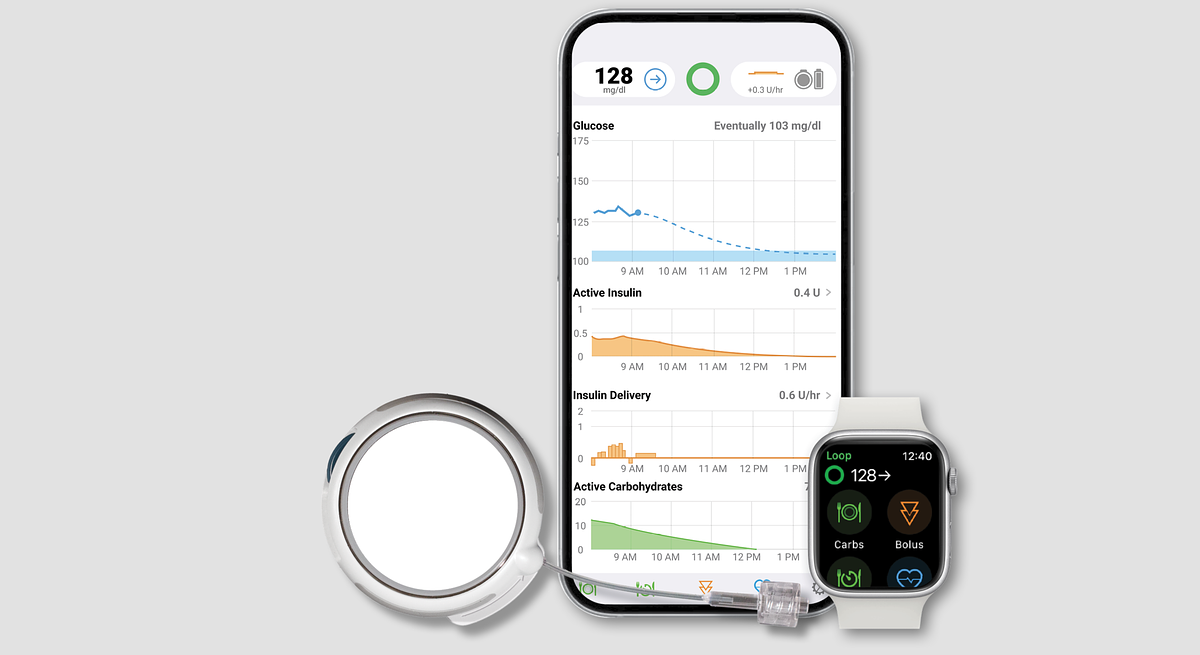
Diabetes Management Enters New Era with Year-Long CGM and Automated Insulin System
MANCHESTER, NH & GERMANTOWN, MD – February 19, 2026 – For millions of people living with type 1 diabetes, the daily routine of device management is a constant, demanding presence. Today, that routine may be set for a seismic shift with the full U.S. launch of an integrated system pairing Sequel Med Tech’s twiist™ Automated Insulin Delivery (AID) System with Senseonics’ Eversense® 365 Continuous Glucose Monitoring (CGM) system.
This combination marks a significant milestone in diabetes care, bringing together the world's only one-year implantable CGM with an advanced, algorithm-powered insulin pump. The collaboration between Sequel and Senseonics promises to dramatically reduce the burden of daily diabetes management, offering a level of convenience and long-term stability previously unavailable.
“Diabetes doesn’t take days off, so the technology I use can’t either,” said Paris Hicks, a person living with type 1 diabetes, in a statement. “Using twiist with Eversense 365 means fewer disruptions, more confidence, and a system that actually works for the way I live. I really appreciate how easy managing my diabetes has been with this combination.”
A New Horizon in Patient Convenience
The most transformative aspect of this new system is the longevity of the Eversense 365 sensor. While most CGMs on the market require users to change sensors every 10 to 15 days, the Eversense 365 is a small sensor implanted under the skin by a healthcare provider that provides accurate glucose readings for a full year. This drastically cuts down on the physical and mental load of frequent site changes, data interruptions, and supply management.
Paired with this long-term monitor is the twiist AID system, which received FDA clearance in March 2024. The system automates insulin delivery using the highly regarded Tidepool Loop algorithm, which originated from the patient-led, do-it-yourself (DIY) diabetes community. The twiist pump continuously adjusts insulin delivery based on the Eversense CGM's real-time data, helping to prevent both high and low blood sugar events with minimal user intervention.
Early feedback from the diabetes community highlights the potential quality-of-life improvements. Some early adopters have reported achieving exceptional time-in-range—a key metric for glucose control—often exceeding 85% within their first week. Many describe a profound sense of relief and improved mental health from the system's reliable automation. However, the transition isn't without a learning curve. Some users have noted that the process for changing the pump's insulin cassette and infusion set is more complex than with other systems, and that the infusion set's adhesive may require extra reinforcement.
Challenging the Status Quo with Choice and Interoperability
Beyond its technical capabilities, Sequel Med Tech is making a strategic statement about the future of diabetes technology. In an industry often defined by proprietary “walled gardens” that lock users into a single company’s ecosystem, Sequel is championing patient choice. The twiist system is also compatible with Abbott's FreeStyle Libre 3 Plus sensor, allowing users and their doctors to select the CGM that best fits their lifestyle and needs.
This commitment to interoperability is deeply connected to the system’s software. By partnering with Tidepool, a nonprofit organization, to power the twiist system, Sequel has embraced an open-source ethos. Tidepool famously took the patient-created Loop algorithm and navigated the rigorous FDA approval process, making it the first FDA-cleared glycemic controller that began as a community-driven initiative.
“People deserve choice and technology that fits the realities of their lives,” said Joanna Mitri, M.D., M.S., Chief Medical Officer at Sequel. “Our goal is to provide a system that supports the many different ways individuals and care teams approach diabetes every day.” This philosophy directly challenges competitors and signals a move toward a more open, patient-centric market where devices from different manufacturers can work together seamlessly.
A Strategic Alliance in a Competitive Market
The partnership between Sequel Med Tech and Senseonics represents a powerful alliance aimed at disrupting a market dominated by established players like Insulet, Tandem Diabetes Care, and Medtronic. While competitors offer sophisticated AID systems, none can match the one-year wear time of the Eversense 365 CGM, which received its own FDA clearance in September 2024.
This unique combination gives the duo a compelling competitive advantage centered on long-term convenience. “National availability of Eversense 365 with twiist marks an important next phase of our journey,” said Brian Hansen, Chief Commercial Officer of Senseonics. “By pairing this groundbreaking CGM with an advanced insulin delivery system, users can spend less time managing devices and more time focusing on their lives.”
The twiist pump also brings its own innovations. Its proprietary iiSure™ Technology directly measures the volume of insulin delivered with every microdose, a feature designed to ensure accuracy and detect potential blockages up to nine times faster than some competing pumps. Furthermore, twiist is the first FDA-approved AID system that can be controlled directly from an Apple Watch, adding another layer of discretion and convenience.
Navigating Real-World Access and Adoption
For any new medical technology, widespread adoption hinges on accessibility and affordability. Sequel Med Tech has stated that the twiist system will be available through the pharmacy channel, which typically simplifies the prescription and fulfillment process compared to the durable medical equipment (DME) pathway. The company is aiming for broad commercial insurance coverage, reporting that 70% of commercial providers are already covering the device.
To ease the financial burden, a patient access program aims to cap out-of-pocket costs at $0 for the first month and no more than $50 per month for refill kits for most commercially insured individuals, though those with high-deductible plans may face higher initial costs. Senseonics and its commercial partner, Ascensia Diabetes Care, are working in parallel to ensure a smooth transition to broad insurance coverage for the Eversense 365 system.
As this integrated solution becomes available nationwide, its ultimate success will be measured not just in technical performance, but in its real-world impact. By offering a year of uninterrupted monitoring paired with intelligent automation and a commitment to patient choice, Sequel and Senseonics are betting that the desire for a less burdensome life with diabetes will drive a new wave of adoption.
