Zeto's New EEG System Aims to End Brain Scan Bottlenecks
- 80% lower access to specialized neurological care in rural/underserved areas vs. urban centers
- 2.5-hour recordings supported by Zeto's New Wave™ system
- $38M+ raised by Zeto, including a $31M Series B round in 2025
Experts would likely conclude that Zeto's New Wave™ system represents a significant advancement in outpatient EEG accessibility, addressing critical staffing shortages and diagnostic delays through its streamlined, user-friendly design and cloud-based analytics.
Zeto's New EEG System Aims to End Brain Scan Bottlenecks
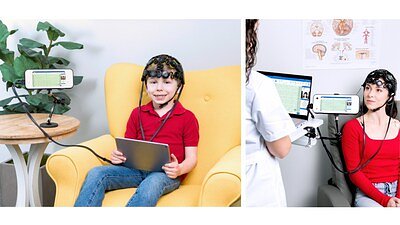
RALEIGH, NC – April 02, 2026 – Zeto, Inc., a medical technology company aiming to revolutionize brain health diagnostics, has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its Zeto New Wave™ system. The announcement marks a significant step forward in the effort to make electroencephalogram (EEG) testing—a critical tool for diagnosing neurological conditions—more accessible and efficient in outpatient clinics and homes.
The Diagnostic Dilemma: Tackling Wait Times and Staff Shortages
For countless patients across the United States, obtaining a timely neurological diagnosis is a race against the clock, but one often stalled by significant logistical hurdles. Patients can wait weeks, sometimes months, for an EEG appointment, a delay that can have profound consequences for conditions like epilepsy, where prompt diagnosis and treatment are crucial. This diagnostic bottleneck is not just an inconvenience; it represents a critical gap in the healthcare system.
The problem is twofold. Firstly, there is a widespread and persistent shortage of specialized EEG technologists. These highly trained professionals are essential for operating traditional EEG equipment, which is often complex and cumbersome to set up. The scarcity of technologists strains clinic resources, limits the number of available appointments, and creates a backlog of patients awaiting vital tests. This challenge is particularly acute in rural and underserved areas, where access to specialized neurological care can be up to 80% lower than in urban centers.
Secondly, the conventional EEG setup process itself is time-consuming. It involves meticulously measuring the patient's head, marking electrode locations, abrading the scalp, and applying a conductive paste or gel to affix the electrodes. This process can be uncomfortable for the patient and requires significant time from a skilled technician, further limiting a clinic's daily capacity. Together, these factors create a challenging environment where both patients and providers are under pressure, delaying care and potentially impacting long-term outcomes.
A New Wave in Neurodiagnostics
Zeto's New Wave™ system is engineered to directly confront these long-standing challenges. Designed for short-term recordings of up to 2.5 hours, the device offers a complete, user-friendly solution for routine outpatient EEGs. Its core innovation lies in its full-coverage headset, available in both adult and pediatric sizes, which features 21 soft-tip, no-mess electrodes. These electrodes are pre-positioned according to the international 10-20 system, a standard for clinical EEG, eliminating the need for manual measurement and marking.
This design dramatically streamlines the setup process. The company claims that healthcare professionals with minimal training, not just specialized EEG technologists, can apply the headset and begin a recording in minutes. By simplifying the procedure and reducing reliance on a limited pool of specialists, clinics can significantly increase their patient throughput and address staffing gaps.
The system is more than just a headset. It is paired with a compact recording device with a built-in display and controls, and it captures synchronized video and audio—essential for correlating brain activity with physical symptoms. The platform is also capable of recording optional signals like ECG, EOG, and EMG, providing a more comprehensive diagnostic picture. Data is streamed to the Zeto Cloud platform, which allows neurologists to access and interpret the EEG data remotely from any location, a feature that further extends the reach of expert care. The cloud platform also supports AI-powered analytics, including seizure detection and seizure burden analysis, helping clinicians to more efficiently parse through data and make decisions.
Navigating a Competitive and Evolving Market
Zeto is not alone in the push to modernize EEG technology. The neurodiagnostics market is active, with established players like Natus Medical offering their own ambulatory and point-of-care EEG systems, such as the Arc Apollo+ for in-home studies and the Natus BrainWatch for rapid seizure detection in emergency settings. The industry is clearly trending toward more portable, wireless, and AI-integrated solutions that can move testing out of the traditional hospital lab and closer to the patient.
Where Zeto appears to be carving its niche is in its holistic approach to the outpatient workflow. Rather than just creating a portable device, the company has built an end-to-end system—from the easy-to-apply headset to the remote cloud interpretation—specifically tailored for physician practices. This focus on usability for non-specialists is a key differentiator in addressing the critical technologist shortage. While some competitors focus on long-term home monitoring or emergency room applications, New Wave™ is optimized for the high-volume, routine EEGs that form the backbone of outpatient neurology.
Zeto's Strategic Ascent in Neurotechnology
The clearance of Zeto New Wave™ is the latest milestone in a deliberate and consistent strategy for the Raleigh-based company. Founded in 2014, Zeto has been on a mission to make brain health insights as common and accessible as other vital signs like blood pressure or heart rate. This is the company's third FDA-cleared device, demonstrating a mature and effective product development pipeline. Its first-generation Zeto WR19 headset was cleared in 2018, followed by the Zeto ONE™ in 2024, a system designed for critical care settings like ICUs and emergency departments.
With the addition of New Wave™, Zeto now offers a comprehensive portfolio targeting distinct clinical environments, from emergency and critical care to outpatient clinics and home use. This product diversification is supported by significant investor confidence. The company has raised over $38 million in funding, including a pivotal $31 million Series B round announced in early 2025, which is fueling its commercial expansion and research efforts.
"New Wave™ is our newest outpatient EEG system, built on lessons learned from our first-generation device, the Zeto WR19," said Florian Strelzyk, PhD, CEO of Zeto, in the company's official announcement. "We listened to neurologists' needs and designed a system that helps clinics expand access to EEG, address staffing challenges, and improve patient experience... Now, providers can choose which system is best for their needs and budget."
By combining user-friendly hardware with a powerful cloud and AI backend, Zeto is positioning itself not just as a device manufacturer, but as a comprehensive neurodiagnostics platform. Its continued innovation and strategic focus on accessibility suggest a determined effort to fundamentally change how and where brain activity is monitored, moving it firmly into the mainstream of everyday medical practice.
