SonoVascular Secures First Close of $6M Series A to Power U.S. Clot-Busting Push
- Series A First Close ($6M Target): SonoVascular secures the first close of its Series A financing, with a target raise of $6 million, to advance its clot-busting technology in the U.S.
- 100% Clot Reduction: First-In-Human trials achieved complete clot removal in all 10 patients without adverse events.
- 900,000 Annual Cases: Venous thromboembolism (VTE) affects 900,000 people in the U.S. yearly, making it the third leading cause of cardiovascular death.
Experts view SonoVascular's SonoThrombectomy™ System as a groundbreaking advancement in VTE treatment, offering complete clot removal without the typical risks of bleeding or vessel damage, and potentially redefining the standard of care.
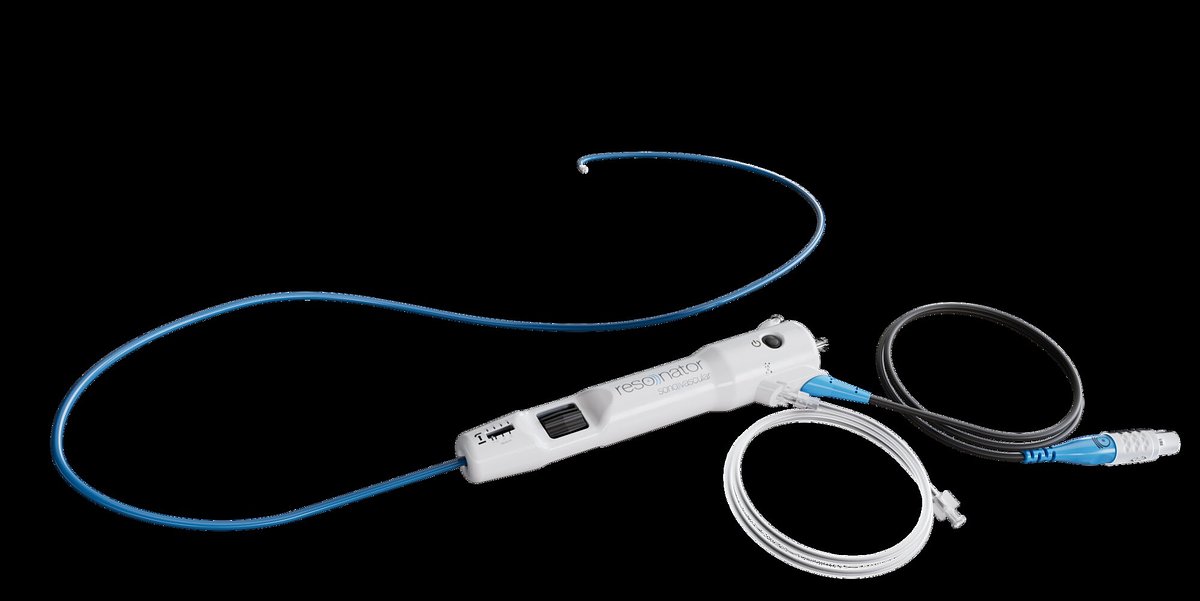
SonoVascular's Clot-Busting Tech Nabs $6M for U.S. Push
CHAPEL HILL, NC – February 03, 2026 – SonoVascular, Inc., a clinical-stage medical device company, announced today it has completed the first close of a $6 million Series A financing round, a major step toward bringing its potentially revolutionary blood clot treatment to the U.S. market. The funding, led by Cary-based Harbright Ventures, will fuel the company's efforts to secure FDA approval for a pivotal U.S. study of its SonoThrombectomy™ System.
The Chapel Hill-based firm is developing a novel approach to treating venous thromboembolism (VTE), a dangerous condition that includes deep vein thrombosis (DVT) and life-threatening pulmonary embolisms (PE). The technology has already demonstrated remarkable success in early human trials, suggesting a new era may be dawning for patients suffering from dangerous blood clots.
A New Frontier in Clot Treatment
VTE affects as many as 900,000 people in the United States each year and is the third leading cause of cardiovascular death. The condition occurs when clots form in deep veins, typically in the legs (DVT), obstructing blood flow. If a piece of the clot breaks free, it can travel to the lungs, causing a pulmonary embolism, which is fatal for 10-30% of individuals within one month of diagnosis.
For years, clinicians have faced a difficult "trade-off" when treating these clots. Current options, including pharmacological drugs (thrombolytics) and mechanical devices that physically remove clots, come with significant risks. High-dose thrombolytics can cause severe bleeding, while mechanical thrombectomy can lead to blood loss and damage to the delicate valves and lining of the veins. This damage can result in Post-Thrombotic Syndrome (PTS), a chronic condition of pain, swelling, and disability that affects up to half of all DVT patients.
SonoVascular's SonoThrombectomy™ System is designed to eliminate this trade-off. It employs a unique mechanism called microbubble-mediated cavitation. The system uses a specialized RESONATOR catheter to deliver a combination of ultrasound energy, microscopic bubbles, and a low dose of the thrombolytic drug tPA directly to the clot. The ultrasound causes the microbubbles to vibrate and implode, a process that mechanically breaks down the clot from the inside out, while the low-dose drug assists in its dissolution. The entire procedure is designed to be completed in a single session in a catheterization lab, with no blood loss or vascular trauma.
"There is currently no ideal device for the treatment of deep vein thrombosis, as existing technologies carry inherent trade-offs, including blood loss and risk of valvular or endothelial injury," said Dr. Lisandro Carnero-Vidal, a vascular surgeon at Clínica La Sagrada Familia in Buenos Aires and the Principal Investigator of the company's first human study.
Flawless Human Trials Signal a Paradigm Shift
The promise of this technology is not merely theoretical. SonoVascular recently completed a First-In-Human (FIH) feasibility study in South America, yielding exceptionally strong results that were presented in part at the 2025 American Venous Forum. The study involved ten patients with DVT, and the outcomes have generated significant excitement in the medical community.
In all ten cases, the SonoThrombectomy System achieved a 100% reduction in clot burden, as verified by an independent core lab using the Marder scoring system. Critically, there were no device-related adverse events reported during the procedures. This level of complete clot removal without complications is a significant achievement.
Perhaps even more importantly, the benefits for patients proved to be lasting. Follow-up examinations at both 30 days and six months post-procedure confirmed significant and sustained improvements in PTS severity scores. Patients reported less pain and swelling, with objective measures like the Villalta and revised Venous Clinical Severity scores showing marked improvement from baseline.
Dr. Carnero-Vidal added, "In this First-In-Human feasibility study, the SonoThrombectomy System demonstrated a favorable safety profile and consistent procedural performance in patients with deep vein thrombosis. While these findings are preliminary, they support further clinical evaluation of intravascular sonication as a therapeutic approach intended to preserve venous valves and endothelium without blood loss."
Strategic Investment Fuels Regulatory Push
The impressive clinical results have attracted significant investor interest. The $6 million Series A first close was led by Harbright Ventures, a firm known for backing disruptive, high-impact technologies. As a sign of its confidence, Harbright's Founding Partner, Robert Ross, has joined SonoVascular's Board of Directors. This financing builds upon previous funding from investors including Carolina Research Ventures and the Wolfpack Investor Network.
"SonoVascular has the rare combination of breakthrough technology and the experienced leadership necessary to redefine the standard of care in VTE," said Ross. "We led this round because SonoVascular is solving the 'trade-off' problem that has plagued DVT treatment for years - achieving total clot reduction without the typical risks of blood loss or vessel damage."
The new capital infusion is earmarked for two critical milestones in 2026. The primary goal is to secure Investigational Device Exemption (IDE) from the U.S. Food and Drug Administration to begin a pivotal clinical trial for peripheral venous applications in the United States. This is a crucial step on the path to commercial approval. Additionally, the company plans to initiate a separate FIH study to evaluate the system's effectiveness in treating pulmonary embolism, expanding its potential impact to the most acute form of VTE.
Easing the Burden on Patients and Hospitals
Beyond the clinical benefits, the SonoThrombectomy System holds the potential for significant economic and logistical advantages for the healthcare system. The annual cost of treating VTE in the U.S. is estimated to be in the billions, ranging from $4.9 to $7.5 billion for DVT and climbing as high as $19.8 billion for PE. By providing a safe, effective, single-session treatment, SonoVascular aims to reduce these costs substantially.
A key part of this value proposition is the potential to avoid lengthy and expensive stays in the Intensive Care Unit (ICU). Many complex VTE cases currently require ICU monitoring, placing a strain on hospital resources.
"With exceptionally strong FIH results, we believe this system is well-positioned to become the new standard of care," Ross continued. "Equally important, SonoVascular's solution will ease the burden on ICU capacity, saving hospitals and patients time and money."
For the hundreds of thousands of patients diagnosed with VTE each year, the development represents a beacon of hope. A treatment that can completely eliminate a dangerous clot in one session, without the risk of bleeding or long-term debilitating syndromes, could transform not just their immediate prognosis but their long-term quality of life. As SonoVascular moves toward its U.S. clinical trials, the medical world will be watching closely to see if this promising technology can deliver on its potential to redefine the fight against blood clots.
