Sesen's AI Workspace Targets Global Life Sciences Compliance Chaos
- 6.5% CAGR: The life sciences translation market is projected to grow at a compound annual rate of over 6.5% through 2035. - Hybrid Model: Sesen's AI Workspace combines AI translation with human review by professional medical linguists. - Regulatory Guardrails: FDA and EMA published principles for good AI practice in January 2026, emphasizing traceability and human governance.
Experts would likely conclude that Sesen's AI Workspace offers a balanced approach to global life sciences compliance by integrating AI efficiency with essential human oversight, addressing critical industry pain points while aligning with regulatory expectations.
Sesen Unveils AI Workspace to Streamline Global Medical Labeling
BOSTON, MA – May 07, 2026 – Sesen, a specialist in life sciences language services, today launched its AI-enabled Labeling Workspace, a new platform designed to tackle the immense complexity of managing regulated medical content for global markets. The centralized environment aims to help pharmaceutical, biotech, and medical device companies streamline the translation and approval of critical documents like drug labels and instructions for use, a process notoriously fraught with risk and inefficiency.
The platform is powered by SesenGPT, a proprietary hybrid translation system that combines artificial intelligence with review by professional medical linguists. This launch enters a market where the life sciences industry is actively seeking to leverage AI's power but remains cautious due to stringent regulatory demands and the high stakes of patient safety.
A High-Stakes Race for Global Compliance
The push to bring new drugs and medical devices to a global market has created a significant bottleneck for life sciences companies: multilingual content management. Every new market requires that labels, patient information leaflets, and regulatory submissions be translated with absolute precision, adhering to a complex and often shifting web of rules from bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
Traditionally, this process has been managed through a disjointed combination of emails, spreadsheets, and manual file transfers. This fragmented workflow often leads to version control nightmares, inconsistent terminology, and a lack of clear audit trails, increasing the risk of costly delays and compliance failures. The industry has been grappling with these operational inefficiencies for years, recognizing that even minor translation errors can have severe consequences for patient safety and lead to product recalls.
Against this backdrop, the market for life sciences translation is experiencing robust growth, with a projected CAGR of over 6.5% through 2035. Companies are increasingly turning to technology to gain a competitive edge, but the unique demands of the sector mean that generic solutions often fall short.
Beyond Automation: A Hybrid Model for Trust
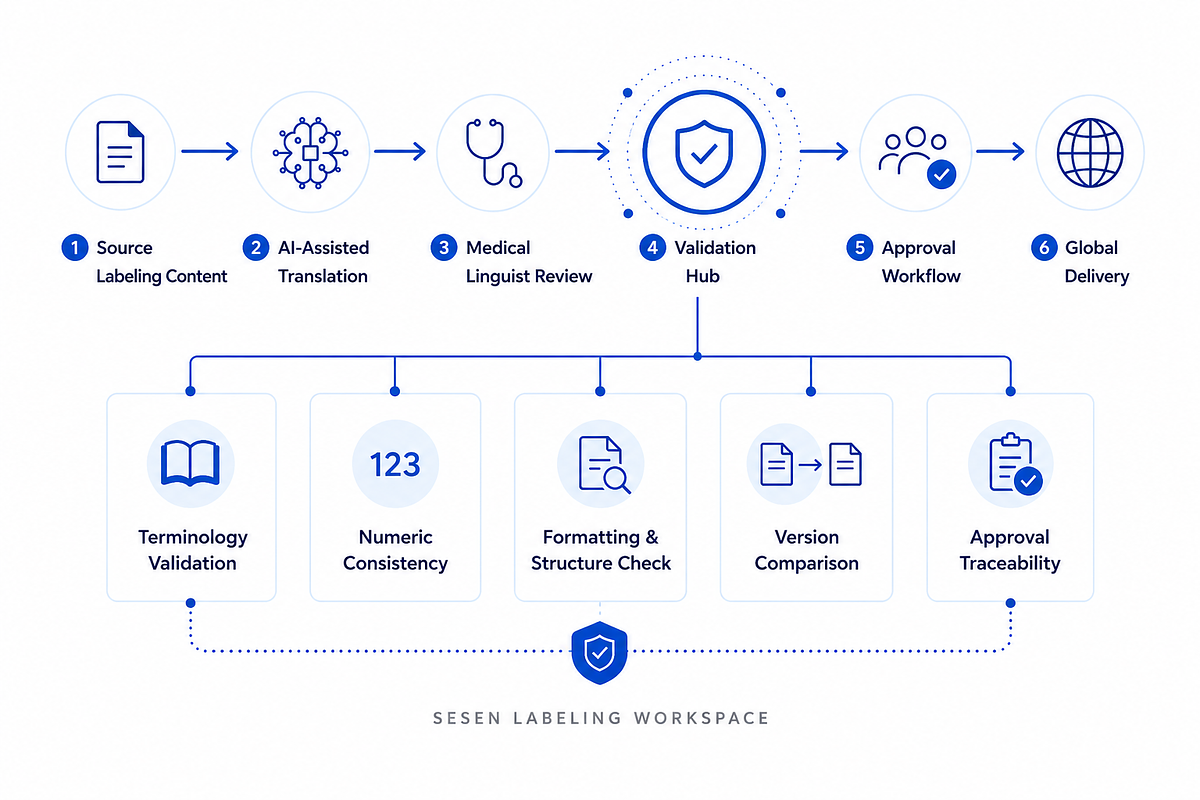
Sesen's new platform aims to address these challenges with a "hybrid" approach that directly confronts the industry's skepticism toward pure AI. Unlike standalone machine translation tools, the Labeling Workspace integrates its AI engine, SesenGPT, into a controlled workflow that maintains human oversight at its core.
The system is designed to use AI for initial translation and to leverage a company's previously approved translations and terminology, ensuring consistency across recurring updates. However, every piece of content is then subject to review and validation by professional native medical linguists. This human-in-the-loop model is critical in a field where context, nuance, and domain-specific knowledge are non-negotiable. It seeks to blend the speed of AI with the precision and reliability of human expertise.
At the heart of this process is the platform's Validation Hub, an AI-driven governance engine. It automatically performs quality assurance checks, verifying terminology against approved glossaries, ensuring numeric consistency, and validating formatting and structure. This automated validation layer is designed to reduce the significant manual QA burden on internal teams while creating a traceable, audit-ready record of every change and approval.
Centralizing Workflows to Unlock Efficiency
The Sesen Labeling Workspace is engineered to serve as a single source of truth for a company's global labeling operations. By centralizing translation, validation, review, and delivery within one environment, the platform aims to eliminate the information silos that plague traditional workflows.
Regulatory, labeling, localization, medical, and quality stakeholders can collaborate within the workspace, providing feedback and approvals in a structured and traceable manner. The system supports a wide range of content, from updates to a Core Company Data Sheet (CCDS) and Summary of Product Characteristics (SmPC) to Instructions for Use (IFU) and post-approval labeling changes.
"The life sciences industry is looking for practical ways to use AI without losing the control required for regulated content,” said Shunee Yee, CEO of Sesen, in the announcement. “The Sesen Labeling Workspace brings AI-assisted translation, expert human review, and validation workflows together in a structured environment designed for real-world labeling operations. Our goal is to help clients improve speed and consistency while maintaining the oversight, traceability, consistency, and quality required for regulated global content.”
Navigating the Cautious Embrace of AI
Sesen’s launch comes at a pivotal moment for AI in highly regulated sectors. While the potential for AI to accelerate drug development and streamline operations is immense, adoption has been cautious. Recent industry analyses have highlighted high failure rates for AI pilot projects, often due to poor data quality, a lack of AI-ready infrastructure, and a disconnect between generic AI tools and the specific validation and compliance requirements of the life sciences.
Regulatory bodies are also moving to provide clearer guardrails. In January 2026, the FDA and EMA jointly published principles for good AI practice, emphasizing the need for traceability, data quality, and robust lifecycle oversight. The clear message is that any AI tool used in this space must be transparent, reliable, and subject to rigorous human governance.
By building its platform around a validation-driven framework and emphasizing human oversight, Sesen appears to be directly addressing these regulatory concerns. The company is also launching pilot programs, allowing potential clients to test the Labeling Workspace with their own content to measure improvements in workflow visibility, turnaround times, and overall multilingual consistency in a real-world setting.
📝 This article is still being updated
Are you a relevant expert who could contribute your opinion or insights to this article? We'd love to hear from you. We will give you full credit for your contribution.
Contribute Your Expertise →