Olympus Launches Advanced Biopsy Needle for Cancer Diagnostics
- 19G and 22G needle sizes available, constructed from Nitinol for enhanced durability and precision.
- Designed to obtain larger, more intact tissue samples, improving diagnostic accuracy for pancreatic cancer and other complex diseases.
- Launch follows successful commercialization in Europe and Japan in fall 2025, now cleared for U.S. market.
Experts in gastroenterology and oncology likely view the SecureFlex™ needle as a significant advancement in EUS-FNB technology, enhancing diagnostic precision and supporting personalized cancer treatment approaches.
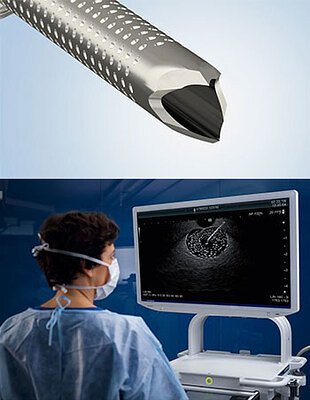
Olympus Launches Advanced Biopsy Needle to Refine Cancer Diagnostics
CENTER VALLEY, PA – January 28, 2026 – Global medical technology leader Olympus announced today the U.S. launch of its SecureFlex™ Single-Use Fine Needle Biopsy (FNB) device, a sophisticated new tool designed to enhance the diagnosis of complex diseases, including pancreatic cancer. The launch marks a significant step forward in the field of endoscopic ultrasound-guided tissue sampling, a critical procedure for accessing and analyzing hard-to-reach lesions within the body.
The SecureFlex™ needle is engineered to overcome common challenges in biopsy procedures, promising physicians greater precision and the ability to obtain larger, more intact tissue samples. This capability is increasingly vital as medicine shifts toward personalized treatments that rely on detailed molecular analysis of tumor tissue.
A Technological Leap in Biopsy Precision
Endoscopic Ultrasound-guided Fine Needle Biopsy (EUS-FNB) is a minimally invasive technique that combines a flexible, lighted tube (endoscope) with ultrasound imaging. This allows physicians to see organs deep within the body, such as the pancreas, and guide a very fine needle through the gastrointestinal wall to collect tissue samples from suspicious lesions. The quality of these samples is paramount for an accurate diagnosis.
The new device introduces several key technological advancements. Its centerpiece is the Dual-Beveled Raptor™ Tip, which features a multi-dimensional cutting surface. An outer edge is designed for smooth puncture, while an inner cutting surface works to guide more tissue into the needle. According to Olympus, this design helps preserve the cellular architecture of the sample, providing pathologists with a clearer and more complete picture for analysis.
"Gastroenterologists require precision and reliability when performing a biopsy for diagnostic accuracy," said Christian Hagie, Vice President of the GI EndoTherapy Business Unit at Olympus Corporation of the Americas, in the company's official announcement. "The SecureFlex EUS-FNB needle offers an advanced design to help physicians perform efficient procedures by facilitating access to complex anatomical structures and obtain adequate samples for diagnostic testing, even in challenging cases."
Further enhancing its performance, the 19G and 22G versions of the needle are constructed from Nitinol, a nickel-titanium alloy known for its shape-memory and super-elastic properties. This construction helps the needle resist bending or deforming as it navigates the body's tortuous pathways, ensuring it remains straight and effective even after multiple passes. For improved real-time guidance, the needle also features a houndstooth-patterned dimpling, which enhances its visibility on the ultrasound monitor, allowing for more confident and precise placement.
A New Front in the War on Pancreatic Cancer
The introduction of the SecureFlex™ needle is particularly timely given the evolution in cancer treatment. For aggressive diseases like pancreatic cancer, the era of one-size-fits-all chemotherapy is giving way to a more nuanced approach involving molecularly targeted therapies and immunotherapies. The success of these modern treatments hinges on identifying specific genetic markers or proteins within a patient's tumor.
Obtaining high-quality core tissue samples—not just loose cells—is essential for this type of comprehensive molecular profiling. The ability to collect larger, structurally intact samples, as the SecureFlex™ is designed to do, directly supports this critical diagnostic need. A more detailed and accurate initial diagnosis can lead to better disease staging and, ultimately, a more effective and personalized treatment plan. This can spare patients from ineffective therapies and guide them toward treatments with a higher probability of success.
By consistently delivering adequate tissue, advanced FNB devices may also reduce the need for repeat biopsy procedures. This not only lowers the risk of complications for the patient, such as infection or bleeding, but also lessens their anxiety and reduces overall healthcare costs.
Navigating a Competitive MedTech Landscape
The launch positions Olympus to strengthen its standing in the highly competitive EUS-FNB device market. The company competes with other MedTech giants like Boston Scientific, Cook Medical, and Medtronic, all of which have developed their own innovative needle designs, such as Franseen and fork-tip needles, to improve diagnostic yield. The industry has seen a clear trend away from older Fine Needle Aspiration (FNA) technology toward FNB devices, as multiple studies and meta-analyses have shown FNB needles provide superior diagnostic accuracy and require fewer passes to obtain a sufficient sample.
The SecureFlex™ is Olympus's answer to this market demand, an advanced offering designed to compete on precision, handling, and sample quality. The device is part of the company's broader strategy to offer a comprehensive ecosystem of gastroenterology solutions. It is designed to work seamlessly with other Olympus products, such as the EU-ME3 Ultrasound Processor, creating an integrated system that supports physicians from imaging to intervention. This strategic move not only aims to capture market share but also reinforces Olympus's identity as a key innovator in the minimally invasive diagnostic and therapeutic space.
From Lab to Clinic: The Path to Market
Following its commercial launch in Europe and Japan in the fall of 2025, the SecureFlex™ device has now received the necessary regulatory clearance for its U.S. debut. To support the rollout, Olympus will be highlighting the new needle at major upcoming medical conferences, including the Cedars-Sinai Endoscopy Symposium in Los Angeles and Orlando Live Endoscopy 2026. These events serve as a critical platform for introducing the technology directly to the gastroenterologists and oncologists who will ultimately use it to improve patient care.
