KROMATID to Debut Platform Setting New Gene Therapy Safety Standard
- 20 kilobases: KROMATID’s platform can detect structural variations as small as 20 kilobases, a two-order-of-magnitude increase over traditional methods.
- 25%+ CAGR: The cell and gene therapy quality control market is valued in the billions and projected to grow at a compound annual rate of over 25%.
- May 2026: KROMATID will unveil its platform at the American Society of Gene & Cell Therapy (ASGCT) Annual Meeting.
Experts in gene therapy and genomics are likely to view KROMATID’s platform as a significant advancement in ensuring genomic integrity, offering a more comprehensive and data-driven approach to safety assessment in cell and gene therapy development.
KROMATID to Debut Platform Setting New Gene Therapy Safety Standard
BOULDER, Colo. – April 30, 2026 – As the field of gene and cell therapy pushes the boundaries of medicine, ensuring the safety and stability of these complex treatments remains a paramount challenge. KROMATID, a Colorado-based genomics company, today announced a significant step toward addressing this challenge, revealing plans to present a breakthrough in genomic integrity assessment at the upcoming American Society of Gene & Cell Therapy (ASGCT) Annual Meeting in May.
The company will unveil a proprietary method for establishing chromosome-scale benchmarks, a move it claims is the cornerstone of the world's first “genomic intelligence platform.” This new category of tool promises to shift the industry away from fragmented safety checks toward an integrated, data-driven framework, potentially transforming how new therapies are developed, approved, and delivered to patients.
A New Benchmark for Genomic Safety
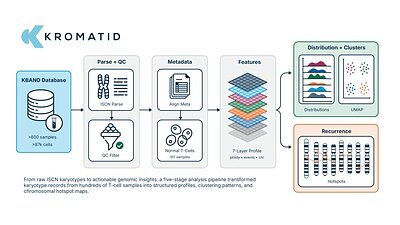
At the heart of KROMATID’s announcement is a solution to a problem that has vexed therapy developers: how to accurately interpret the genomic changes that can occur during the manufacturing of gene-edited cells. Current methods for assessing genomic integrity often provide an incomplete picture. Traditional G-banding karyotyping, a long-standing tool for visualizing chromosomes, is limited in its resolution, typically only detecting large-scale changes of around 10 megabases. This leaves a critical blind spot for smaller, yet potentially significant, structural variations.
More modern techniques like Whole Genome Sequencing (WGS) offer base-pair level detail but can struggle to identify and correctly assemble large, complex chromosomal rearrangements. This leaves developers with isolated data points, making it difficult to distinguish between benign cellular variations and potentially dangerous, therapy-induced instability.
KROMATID’s platform, KROMASURE™, tackles this gap with what it calls “high-depth cytogenetic analysis.” By directly visualizing the genomes of thousands of individual cells, the technology can detect structural variations—including translocations, inversions, and insertions—as small as 20 kilobases. This represents a two-order-of-magnitude increase in resolution over traditional methods. More importantly, by analyzing vast datasets, the company is building the first data-driven benchmarks for chromosomal stability. For the first time, developers can compare the genomic profile of their therapeutic cells against a standardized reference, providing crucial context to understand whether an observed variant is a cause for concern or within an acceptable range.
Navigating a Complex Regulatory Landscape
The timing of KROMATID’s announcement is particularly salient. Regulatory bodies, including the U.S. Food and Drug Administration (FDA), are placing increasing emphasis on the comprehensive assessment of genomic integrity for new cell and gene therapies. In fact, the FDA issued new draft guidance as recently as April 2026, recommending advanced methods to evaluate off-target editing and chromosomal alterations.
A key theme in regulatory discussions is the use of “orthogonal methods”—multiple, independent techniques used to validate a single finding. KROMATID's visualization-based assay is positioned as an ideal orthogonal partner to sequencing-based methods. Where sequencing provides the code, the company's platform provides the architectural blueprint, confirming that the chromosomes are structured correctly after editing. This dual-pronged approach provides the robust data package that regulators are seeking and is essential for building confidence in a therapy’s safety profile before it enters human trials.
This enhanced level of clarity has profound business implications. The cell and gene therapy quality control market, valued in the billions and projected to grow at a compound annual rate of over 25%, is fueled by the high stakes of development. By providing clearer, more decisive data early in the process, the platform promises to de-risk a notoriously expensive and lengthy development cycle. Strengthening Investigational New Drug (IND) applications with this level of detailed safety data could reduce regulatory hurdles, accelerate timelines, and secure the massive investments required to bring these therapies to market.
From Fragmented Data to Actionable Intelligence
The vision articulated by KROMATID extends beyond a single assay. The creation of a “genomic intelligence platform” signals a strategic shift from providing isolated tests to delivering integrated, decision-ready insights. This approach seeks to unify disparate streams of genomic data into a single, interpretable framework that supports developers throughout the entire therapeutic lifecycle.
“This is a defining moment for cell and gene therapy,” said Terry Opgenorth, CEO of KROMATID, in the company’s press release. “What KROMATID is building - a genomic intelligence platform – which brings clarity, context, and confidence to a space that has long operated without it. This realizes KROMATID's vision to de-risk cell and gene therapy development and enable more therapies to get to market.”
This platform model aims to empower developers from the earliest stages of discovery, helping them select the most stable cell lines and editing strategies. It continues through preclinical development, providing the rigorous safety data needed for regulatory filings. Finally, it extends to manufacturing and batch release, ensuring that the final product administered to patients meets the highest standards of quality and genomic integrity.
The Broader Impact on Precision Medicine
While the immediate impact of KROMATID’s technology is focused on safety and regulatory efficiency, its long-term potential lies in enabling the next generation of more complex and ambitious therapies. As scientists develop treatments that involve multiple gene edits or more intricate cellular engineering, the need for a foundational guarantee of genomic stability becomes non-negotiable.
By establishing a new, higher standard for quality control, such platforms provide the confidence needed for the entire field to advance. This foundational safety layer allows researchers to innovate more boldly, knowing they have the tools to monitor and control for unintended genomic consequences. Ultimately, this leads to a future where precision medicine is not only more powerful but also demonstrably safer for the patients who depend on it. The move from simple genetic assessments to a comprehensive intelligence framework may prove to be a critical inflection point in the maturation of gene therapy from a promising science to a standard of medical care.
📝 This article is still being updated
Are you a relevant expert who could contribute your opinion or insights to this article? We'd love to hear from you. We will give you full credit for your contribution.
Contribute Your Expertise →