Ellipsys System Returns, Offering Less Invasive Dialysis Access Option
- 95-100%: Technical success rates of the Ellipsys system
- 96%: One-year secondary patency rate for dialysis access
- 2026: Planned relaunch year of the Ellipsys system
Experts view the Ellipsys system as a significant advancement in dialysis access, offering a minimally invasive alternative to traditional surgery with strong clinical success rates and potential to improve patient outcomes.
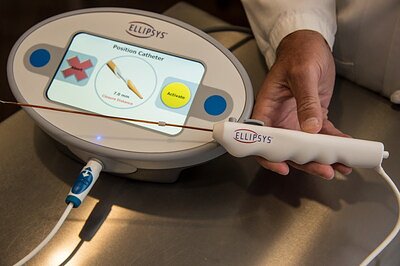
Ellipsys System Set for 2026 Relaunch, Promising a Less Invasive Future for Dialysis Patients
SAN CLEMENTE, CA – April 02, 2026 – A groundbreaking technology that allows for the creation of dialysis access without open surgery is poised to make a significant return to the U.S. market. Ellipsys Medical, Inc. has announced the planned reintroduction of its Ellipsys® Vascular Access System, with a phased rollout anticipated to begin in mid-2026. The announcement signals a new chapter for the minimally invasive device and offers renewed hope for thousands of patients with end-stage renal disease who rely on hemodialysis to live.
For decades, the standard for creating an arteriovenous (AV) fistula—the vascular “lifeline” required for effective dialysis—has been a surgical procedure. The return of Ellipsys, which had been in a period of limited market availability, directly addresses a growing demand from clinicians and patients for less invasive, more patient-friendly alternatives.
A New Lifeline Without the Knife
The core innovation of the Ellipsys system is its ability to create a durable AV fistula percutaneously, meaning through the skin. Using a single, ultrasound-guided catheter, a physician can fuse an artery and a vein in the arm to create the high-flow connection necessary for dialysis. The entire procedure typically takes less than 30 minutes, can be performed in a hospital outpatient department, ambulatory surgery center, or office-based lab, and usually requires only local anesthesia.
This approach stands in stark contrast to traditional surgical fistula creation, which involves incisions, vessel dissection, and a longer recovery period. The Ellipsys procedure leaves no surgical scar and requires no permanent implants. For patients, the benefits are profound.
"For patients living with kidney failure, every procedure has a meaningful impact on their daily lives," said Terry Litchfield, MPA, CPC, President of Access Solutions, in the company's press release. "By offering a minimally invasive option that avoids open surgery and supports reliable dialysis access, Ellipsys can help reduce the burden of care and improve the patient experience."
Clinical studies support the system's efficacy, with technical success rates frequently reported between 95% and 100%. While a significant percentage of patients may require a secondary procedure, such as a balloon angioplasty to help the fistula mature, the long-term outcomes are promising. Published data shows one-year secondary patency rates—a measure of how long the access site remains usable with minor interventions—as high as 96%, with some studies demonstrating a two-year patency of over 92%.
A Device's Journey Through the Corporate Maze
The path for the Ellipsys system back to the market has been complex. Originally developed by Avenu Medical, the device received its De Novo clearance from the U.S. Food and Drug Administration (FDA) in 2018, marking a significant milestone in vascular access innovation. Its potential was quickly recognized, leading to the company’s acquisition by medical technology giant Medtronic in 2020.
However, following the acquisition, the device entered what the current announcement describes as a "period of limited market availability." While the specific reasons for the limited rollout under previous ownership are not publicly detailed, the re-emergence of the technology under a new, dedicated entity—Ellipsys Medical, Inc.—suggests a strategic reset. This “phoenix story” points to a renewed and focused effort to make the technology widely accessible.
"We are excited to restore access to Ellipsys for clinicians who have continued to advocate for minimally invasive fistula creation," stated Brian Driscoll, CEO of Ellipsys Medical. "The system has a strong clinical foundation, and its return reflects both the demand we've heard from the field and our commitment to expanding vascular access options."
Re-entering a Competitive and Evolving Market
Ellipsys Medical re-enters a market that is increasingly receptive to innovation but also presents significant challenges. The primary competitor in the percutaneous AV fistula space is the WavelinQ™ EndoAVF System from Becton, Dickinson and Company (BD). While both devices create fistulas without surgery, they employ different mechanisms. WavelinQ uses a dual-catheter system with radiofrequency energy and requires fluoroscopy, exposing the patient to radiation. Ellipsys, by contrast, uses a single catheter with thermal energy and relies solely on ultrasound guidance.
Beyond direct competition, widespread adoption faces several hurdles. Not all patients are anatomically suitable for a percutaneous procedure, requiring specific vessel diameters and proximity. Furthermore, establishing new procedures requires robust training programs to ensure physicians are proficient and comfortable with the technique. Reimbursement policies from insurers can also be slow to adapt to new technologies, potentially limiting initial access.
Ellipsys Medical appears ready to meet these challenges head-on. The company's planned phased reintroduction includes the restoration of physician training programs, clinical education initiatives, and a comprehensive customer support infrastructure. This strategic approach is designed to ensure that as the device becomes available, the ecosystem needed to support its successful adoption is firmly in place.
As healthcare continues its inexorable shift toward less invasive treatments that reduce recovery time and improve patient comfort, the return of the Ellipsys system is a timely development. Its reintroduction not only adds a powerful tool to the arsenal of vascular access specialists but also represents a renewed commitment to advancing the standard of care for a patient population that endures the daily challenges of life with kidney failure.
