Ziptek’s Biolock Aims to Remodel the Tissue Repair Market
- $15B+ global orthopedic soft tissue repair market in 2024, projected to exceed $20B by 2030
- 24% reduction in re-operation rates and 60% faster recovery times reported with Ziptek’s Biolock™
- 500+ surgical cases with up to 4 years of follow-up supporting early data
Experts view Ziptek’s Biolock™ as a promising innovation in tissue repair, with its biologically synchronized healing approach potentially offering stronger, more functional long-term outcomes compared to traditional mechanical fixation methods.
Ziptek’s Biolock Aims to Remodel the Tissue Repair Market
SARASOTA, FL – April 09, 2026 – An emerging medical device company is making significant waves in the multi-billion dollar orthopedic market, challenging long-held surgical principles with a technology designed to work with the body's own healing capabilities rather than against them. Sarasota-based Ziptek has announced major clinical and commercial milestones for its ZipE® Biolock™ technology, a novel tissue repair system that could represent a paradigm shift in how surgeons approach everything from common rotator cuff tears to complex trauma.
Unlike traditional methods that rely on tightly cinched sutures and rigid anchors to mechanically hold tissue in place, Ziptek’s system introduces a new philosophy: biologically synchronized healing. The company's recent progress, including key institutional adoption and growing international interest, suggests the medical community is taking notice.
A New Paradigm in Tissue Repair
The fundamental challenge in orthopedic repair is that biological tissue and mechanical fixation devices heal on different timelines. Conventional sutures can create high-pressure points that lead to what surgeons call tissue strangulation, tearing, or gapping, where the repaired tendon pulls away from the bone. This can lead to a repair that heals with weak scar tissue or fails altogether.
Ziptek’s ZipE® Biolock™ was engineered to solve this problem. The system is a resorbable device, meaning it is naturally absorbed by the body over time. Its unique design provides what the company calls “controlled tendon compression,” a feature that distinguishes it from all existing anchor-based technologies. Instead of a high-pressure loop, the Biolock device distributes force over a wider surface area, creating a more stable and tissue-friendly environment.
"Current technologies can negatively impact healing due to mechanical limitations, including tissue strangulation, tearing, and gapping caused by traditional suture loops," a Ziptek spokesperson stated in a recent press release. "The ZipE® Biolock™ changes the mechanics of repair, creating a more tissue-friendly environment."
This approach allows for progressive fixation release as the device is resorbed, synchronizing the mechanical support with the body’s natural tissue regeneration. The goal is to facilitate true tissue remodeling, potentially leading to stronger, more functional long-term healing and avoiding the pitfalls of repairs that rely on scar formation.
Navigating a Market of Giants
Ziptek is stepping into an arena dominated by corporate titans like Stryker, Smith & Nephew, and Arthrex. The global orthopedic soft tissue repair market was valued at over $15 billion in 2024 and is projected to exceed $20 billion by 2030. The rotator cuff repair segment alone represents a nearly $900 million market. To compete, a new entrant needs more than just a good idea; it needs undeniable clinical and commercial validation.
After launching commercially about a year ago, Ziptek has secured a landmark achievement: product approval and adoption by Tampa General Hospital (TGH) and its parent system, Florida Health Sciences Center. The decision, which followed a review by the hospital's Value Analysis Committee, places the technology alongside products from established industry leaders. The move was championed by Dr. Roy Sanders, President of The Florida Orthopedic Institute, a key figure in advocating for the technology's integration into the TGH system.
This institutional validation has also caught the attention of hospital leadership. Tampa General Hospital CEO John Couris was reportedly so impressed after hearing a pitch from Ziptek that he encouraged the hospital's venture arm to explore a potential investment. For a company that was self-funded by its founder, Dr. William Bennett, until 2014 and was seeking Series A funding as recently as 2018, this level of interest from a major health system's investment division marks a significant turning point. With an open funding round currently underway, Ziptek appears poised to capitalize on this momentum to fuel its next phase of growth.
The Clinical Promise and the Path to Proof
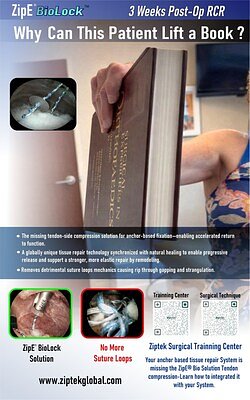
The ultimate measure of any medical device is its impact on patient outcomes. Ziptek reports that its technology is supported by over 500 surgical cases with follow-up periods of up to four years. Early data from this cohort, the company claims, shows drastically reduced re-operation rates compared to published benchmarks for traditional repairs. Affiliated reports even suggest the system may reduce returns to the operating room by 24% and accelerate patient recovery times by up to 60%.
These figures are compelling, particularly in rotator cuff surgery, where re-tear rates can be notoriously high. The image of a patient lifting a book just three weeks after surgery—a recovery timeline significantly shorter than many conventional protocols—paints a powerful picture of the technology's potential. The company has garnered over 28 patents and numerous innovation awards, bolstering its scientific credibility.
However, while the company's internal data and surgeon testimonials are positive, the broader medical community typically awaits independent, peer-reviewed clinical trials published in major medical journals to fully validate such claims. While the FDA granted the device 510(k) clearance as a Class II device back in March 2017, demonstrating its substantial equivalence to existing products, the next step for widespread adoption will be the publication of robust clinical evidence to independently corroborate the impressive results reported by the company and its network of early adopters.
From Local Innovator to Global Contender
Ziptek's strategy extends far beyond its home state of Florida. The company has already secured regulatory approval in the United States, Mexico, and the United Arab Emirates, establishing a foothold in key international markets. This global expansion is supported by active surgeon training programs, with recent sessions held in Mexico City and Dubai in collaboration with regional distribution partners. The company notes that surgeons who previously used systems from major orthopedic manufacturers are increasingly transitioning to the ZipE® Biolock™, suggesting a growing preference for its unique approach.
Further global expansion will require navigating complex regulatory landscapes. Entry into the European Union, for instance, requires adherence to the stringent Medical Device Regulation (MDR), a process that can take 12-18 months for a higher-risk implantable device and demands extensive clinical data. Similarly, markets in Canada and the Asia-Pacific region have their own rigorous requirements, though existing FDA clearance can sometimes streamline these processes.
To continue building its momentum, Ziptek is actively engaging with the orthopedic community. The company is scheduled to showcase its technology at the upcoming Shoulder360 conference in Miami and will host a surgeon-led dinner at the Annual Osteopathic Orthopedic meeting in Orlando. These events provide a platform for surgeons to discuss the technology and for Ziptek to demonstrate why it believes its bio-integrative solution represents the future of tissue repair.
