TheWell Bioscience Launches Medium to Tackle Stem Cell Manufacturing Hurdles
- $10 billion: The hMSC therapeutics market is projected to surpass this value by 2033. - 1,200+ clinical trials: The global pipeline for MSC-based therapies currently includes this many ongoing studies. - Xeno-free formulation: RocketCell™ eliminates all non-human animal components, addressing variability and regulatory concerns.
Experts agree that TheWell Bioscience's RocketCell™ medium represents a significant advancement in stem cell manufacturing, addressing critical regulatory and scalability challenges while preserving cell potency, which is essential for the clinical success of regenerative therapies.
TheWell Bioscience Launches Medium to Tackle Key Stem Cell Manufacturing Hurdles
MONMOUTH JUNCTION, NJ – March 04, 2026 – As the promise of regenerative medicine moves closer to clinical reality, the challenge of manufacturing living cells at scale and with consistent quality has become a critical bottleneck. Addressing this, TheWell Bioscience today announced the launch of RocketCell™ hMSC Xeno-Free Complete Growth Medium, a new product designed to standardize the production of human mesenchymal stem cells (hMSCs).
The chemically defined, animal-component-free medium aims to provide researchers and biomanufacturers with a more reliable and reproducible method for expanding hMSCs, which are central to hundreds of clinical trials for conditions ranging from osteoarthritis to autoimmune disorders.
This launch enters a rapidly growing hMSC therapeutics market, with some projections estimating its value will surpass $10 billion by 2033, driven by a rising prevalence of chronic diseases and an aging global population. However, realizing this potential depends heavily on overcoming fundamental manufacturing challenges.
The Quest to Eliminate Variability
For decades, the workhorse of cell culture has been fetal bovine serum (FBS), an animal-derived cocktail of growth factors. While effective, FBS is a biologically undefined substance, with its composition varying significantly from batch to batch. This variability introduces a major risk for therapeutic development, potentially altering cell behavior and making it difficult to achieve consistent results.
Furthermore, the use of animal-derived components carries the risk of transmitting pathogens, such as viruses or prions, and can trigger immune responses in patients. These risks have drawn increasing scrutiny from regulatory bodies worldwide.
RocketCell™ is engineered to directly address these long-standing issues. Its fully defined, xeno-free formulation eliminates all non-human animal components, replacing the uncertainty of serum with a precise recipe of ingredients. According to TheWell Bioscience, this ensures high-performance cell expansion, allows for efficient growth even at low seeding densities, and maintains the crucial therapeutic properties of the stem cells over long-term culture—a vital requirement for generating clinically relevant cell numbers.
Navigating a Shifting Regulatory Landscape
The move away from animal-derived materials is not just an industry preference but a growing mandate from regulatory agencies. Both the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have issued guidance encouraging, and in many cases requiring, the use of chemically defined and animal-free components for the manufacturing of cell-based therapies. The goal is to enhance product safety, quality, and consistency.
By creating a fully defined system, companies can streamline their regulatory submissions and reduce the burden of demonstrating product safety and comparability. TheWell Bioscience is positioning its new medium as a tool to achieve this “regulatory readiness.”
“The industry is rapidly transitioning toward defined, xeno-free bioprocessing systems,” said John Huang, Ph.D., CEO of TheWell Bioscience, in a statement accompanying the launch. “RocketCell™ hMSC Complete Growth Medium is not only a high-performance xeno-free medium—it is also part of our end-to-end xeno-free 3D cell expansion platform.”
This platform strategy integrates the new medium with the company’s existing synthetic VitroGel® hydrogels and VitroPrime™ culture systems. Dr. Huang stated that this integrated approach is key to “building the foundational 3D cell infrastructure needed to support scalable, reproducible, and clinically translatable cell manufacturing.”
Enhancing Efficiency in a Competitive Market
TheWell Bioscience enters a competitive field where established life science giants like Thermo Fisher Scientific, Lonza, and Sartorius already offer a range of xeno-free and chemically defined media. The market's focus has intensified on solutions that not only meet regulatory standards but also improve the economic feasibility of cell therapy manufacturing.
To stand out, TheWell is highlighting features designed for improved laboratory efficiency and scalability. One notable feature is a “weekend-friendly” workflow. The medium is optimized for an alternate-day feeding schedule, reducing the hands-on labor and resources required to maintain large-scale cultures. This seemingly small convenience can have a significant impact on operating costs and throughput, especially in high-demand manufacturing environments.
Experts in the field note that while the upfront cost of high-quality, chemically defined reagents can be higher than traditional serum, the long-term benefits of reduced batch failure, simplified regulatory processes, and improved consistency often lead to lower overall costs and a faster path to market.
Preserving Potency from Lab to Clinic
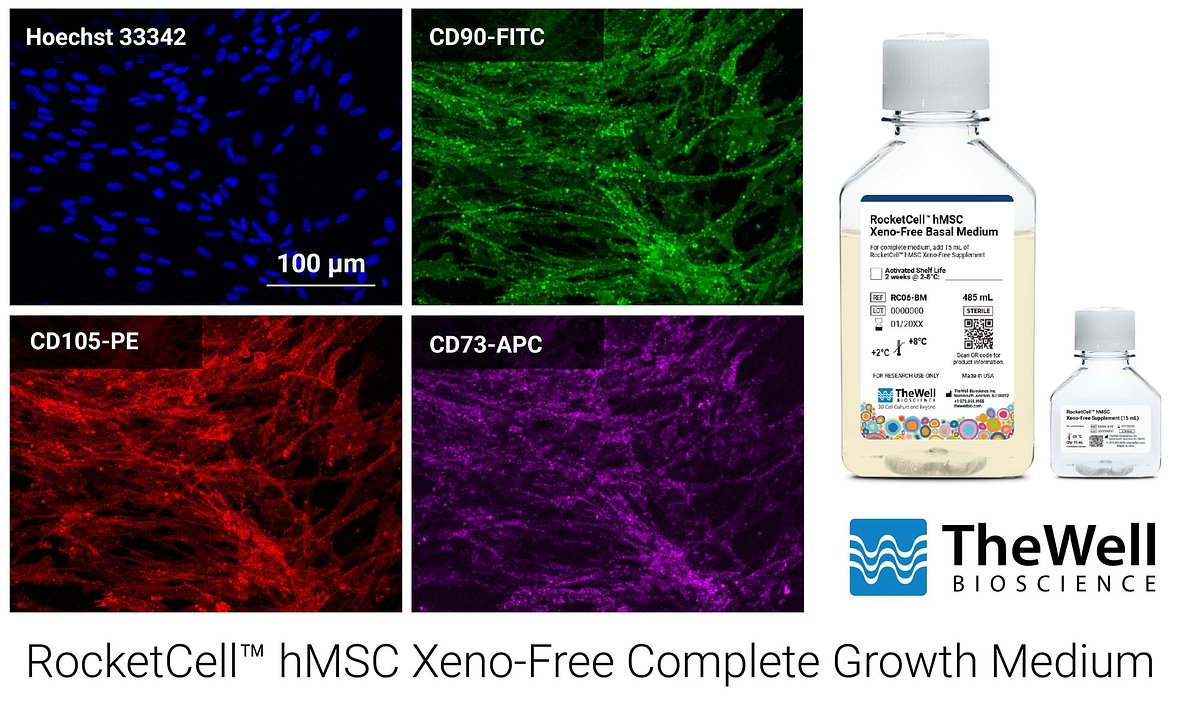
Ultimately, the success of any cell culture medium hinges on its ability to preserve the fundamental biological identity and therapeutic potential of the cells. For hMSCs, this means maintaining their multipotency—the ability to differentiate into other cell types—and their immunomodulatory functions.
The company reports that cells expanded in RocketCell™ medium retain their classical hMSC markers and their sustained capacity for tri-lineage differentiation into bone (osteogenic), cartilage (chondrogenic), and fat (adipogenic) cells, even after extended periods of expansion. This assurance is critical for developers, as it confirms that the scaled-up cell population remains therapeutically functional.
As the global pipeline for MSC-based therapies continues to expand, with over 1,200 clinical trials underway, the demand for robust and standardized manufacturing solutions will only intensify. The introduction of advanced platforms like RocketCell™ represents a crucial step in industrializing cell production, moving these promising treatments from bespoke laboratory processes to reliable, off-the-shelf therapeutic products.
