Skyhawk's Huntington's Drug Gains Fast-Track Status in Australia
- 2,000 Australians and 40,000 symptomatic patients in the U.S. affected by Huntington's disease
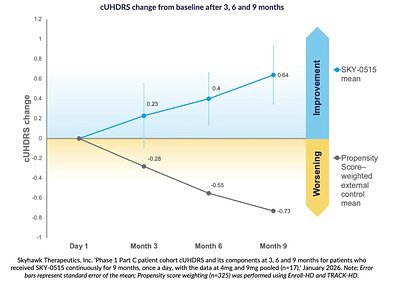
- +0.64 points mean improvement on the cUHDRS scale in early trials (vs. expected -0.73 decline)
- Up to two years faster approval timeline through Australia's provisional pathway
Experts view SKY-0515 as a promising first-in-class treatment with strong potential to modify Huntington's disease progression, though larger trials are needed to confirm efficacy.
Skyhawk's Huntington's Drug Gains Fast-Track Status in Australia
BOSTON, MA – March 02, 2026 – In a development providing a significant new spark of hope for the Huntington's disease community, Skyhawk Therapeutics announced today that its experimental oral drug, SKY-0515, has met the eligibility criteria for a provisional approval pathway in Australia. The country's Therapeutic Goods Administration (TGA) determined that the therapy, a potential first-in-class treatment, is a suitable candidate for accelerated review, a crucial step that could bring a much-needed therapy to patients sooner.
Following the TGA's determination, the Boston-based biotechnology company confirmed it has already submitted its formal application for provisional registration. This move signals a potential shift in the landscape for Huntington's disease, a relentless and fatal neurodegenerative disorder for which there are currently no approved treatments that can slow or halt its progression.
A New Pathway for a Devastating Disease
Huntington's disease is an inherited condition that causes the progressive breakdown of nerve cells in the brain, deteriorating a person's physical and mental abilities over time. It affects hundreds of thousands worldwide, including over 2,000 Australians and 40,000 symptomatic patients in the United States, leaving families to navigate a challenging and ultimately tragic journey.
The TGA's provisional approval pathway is specifically designed for promising new medicines that treat serious or life-threatening conditions with high unmet medical need. By granting eligibility, the regulatory body acknowledges that SKY-0515 shows preliminary evidence of being a major therapeutic advance. This pathway can potentially shorten the time to market by up to two years compared to the standard process, allowing patients earlier access while the company gathers more comprehensive long-term clinical data required for full approval.
"We are grateful that the TGA has determined SKY-0515 for the treatment of Huntington's disease meets its eligibility criteria for potential provisional approval," said Bill Haney, Skyhawk's CEO, in a statement. "This represents an important first step toward what could be an accelerated path to approval in Australia and the world, offering the possibility of a disease modifying treatment for Huntington's patients who urgently need therapies for this devastating disease."
The Science Behind SKY-0515
At the heart of this development is a novel scientific approach. SKY-0515 is an orally administered small molecule designed to modify RNA splicing. It was developed using Skyhawk's proprietary SKYSTAR® platform, which aims to correct the errors in RNA that lead to disease. In the case of Huntington's, the drug targets the underlying genetic cause by reducing the production of mutant huntingtin protein (mHTT), the toxic protein that drives the disease's pathology.
Early clinical data from the Phase 1C portion of Skyhawk's study has been encouraging. In a small, pooled analysis of 17 patients who received the drug daily for nine months, participants showed a mean improvement of +0.64 points on the Composite Unified Huntington's Disease Rating Scale (cUHDRS), a standard measure of motor and cognitive function. This stands in stark contrast to the natural course of the disease, where a worsening of -0.73 points would be expected over the same period, according to historical data from large observational studies.
While this early data is promising, it comes from a small, early-phase trial primarily designed to assess safety and tolerability, which the company reports has been generally good. The true test of SKY-0515's efficacy will come from larger, more robust clinical trials. The company has completed enrollment for its Phase 1C trial, with over 115 patients now on trial with the drug, and is advancing into later-stage studies.
Navigating a Challenging Clinical Landscape
The path to a successful Huntington's disease therapy is notoriously difficult, a reality underscored by past setbacks in the field. The high-profile halting of a late-stage trial for Roche's tominersen in 2021 served as a sobering reminder of the complexities of treating this disease. However, the field has persisted, with several companies pursuing innovative strategies.
SKY-0515's key advantage lies in its design as a once-daily oral pill. This method of administration offers a significant improvement in convenience and accessibility compared to other investigational approaches that require invasive procedures like spinal injections or direct brain infusions. It is part of a competitive new wave of oral small molecules aiming to modify the disease at its source, with companies like PTC Therapeutics also advancing a similar type of therapy through late-stage trials.
Skyhawk's progress represents a validation of its RNA-modulating platform, which has attracted significant investment and major partnerships with pharmaceutical giants like Merck, Biogen, and Genentech for developing treatments across a range of diseases.
A Strategic Bet on Global Access
Skyhawk's decision to pursue the Australian provisional approval pathway is a calculated strategic move. For a clinical-stage biotech, securing an accelerated review in a key market can not only provide early revenue but also build regulatory momentum for submissions in the United States and Europe. With substantial financial backing from top-tier investors and a network of powerful pharmaceutical partners, Skyhawk is well-positioned to fund its ambitious clinical program.
The next and most critical step is the ongoing Phase 2/3 FALCON-HD clinical study. This large-scale, international trial will enroll hundreds of participants across dozens of sites worldwide, including Australia and New Zealand. Its goal is to provide the definitive, long-term safety and efficacy data needed to confirm the promising early signals and secure full regulatory approval globally.
For the thousands of families affected by Huntington's disease, the TGA's decision is more than a regulatory milestone; it is a tangible sign of progress in a fight that has long been starved for victories. While the journey for SKY-0515 is far from over, this accelerated pathway provides a clear and hopeful route forward, moving one step closer to a future where a diagnosis of Huntington's disease is not a death sentence.
