Skyhawk Drug Shows Promise, Reversing Decline in Huntington's Trial
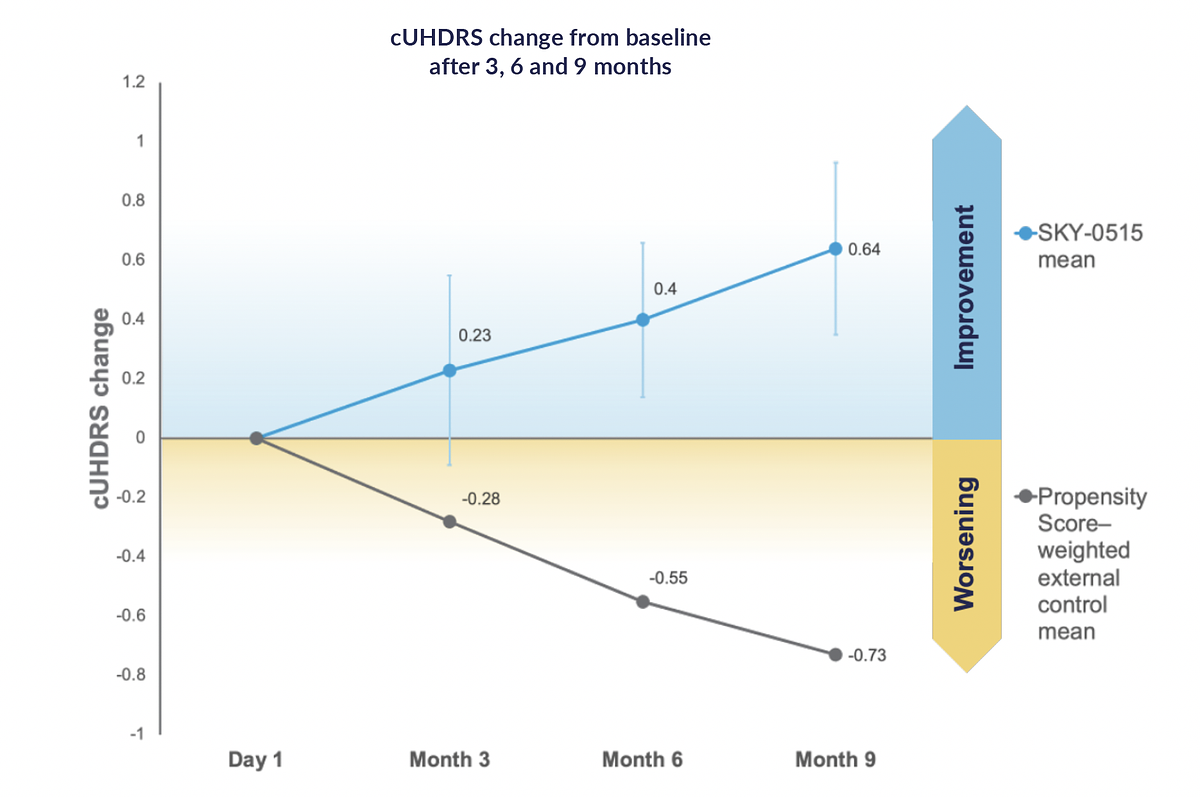
- cUHDRS Improvement: Patients on SKY-0515 showed a mean improvement of +0.64 points over nine months, reversing the expected decline of -0.73 points in Huntington's disease progression.
- mHTT Reduction: The drug achieved a dose-dependent reduction of mutant huntingtin protein (mHTT) by up to 62% in blood levels, the highest demonstrated to date.
- PMS1 Reduction: SKY-0515 reduced mRNA levels of PMS1, a key disease driver, by 26%.
Experts view SKY-0515 as a promising, potentially disease-modifying therapy for Huntington's, highlighting its unique dual mechanism, strong early efficacy data, and favorable safety profile as significant advancements in the field.
Skyhawk Drug Shows Promise, Reversing Decline in Huntington's Trial
BOSTON, MA – January 27, 2026 – In a development offering significant hope for the Huntington's disease community, Skyhawk Therapeutics today announced encouraging interim results from its Phase 1 clinical trial for an investigational oral drug, SKY-0515. The nine-month data shows that patients taking the therapy not only avoided the expected disease progression but demonstrated a measurable improvement in clinical scores, a rare feat in the challenging field of neurodegenerative disease research.
Huntington's disease is a devastating, inherited, and ultimately fatal disorder that progressively breaks down nerve cells in the brain, impacting movement, mood, and cognitive abilities. With over 40,000 symptomatic patients in the U.S. and no approved treatments to slow or halt its progression, the search for a disease-modifying therapy has been relentless and fraught with setbacks. Skyhawk's announcement marks a potentially pivotal moment, suggesting a new path forward may be on the horizon.
A Glimmer of Hope in Clinical Data
The most striking finding from the nine-month interim analysis comes from the Composite Unified Huntington's Disease Rating Scale (cUHDRS), a standard measure used to track the progression of the disease. Patients in the trial receiving SKY-0515 demonstrated a mean improvement from baseline of +0.64 points. This stands in stark contrast to the natural course of the illness, where symptomatic patients are expected to worsen by an average of -0.73 points over the same nine-month period, according to established natural history data.
This divergence from the expected decline is a statistically and clinically significant signal. The comparison was made using a propensity score weighting method against large observational studies, including Enroll-HD and TRACK-HD, to create a robust external control group.
"I am very encouraged by these safety and early efficacy data from SKY-0515's Phase 1 Part C trial in patients, showing divergence in cUHDRS away from expected natural history deterioration at the three, six, and nine month prespecified analyses," stated Ed Wild, Professor of Neurology at University College London, in the company's press release. He added that the drug has been well tolerated at all doses tested. This favorable safety profile is crucial, as many investigational drugs for brain disorders have been sidelined by adverse effects.
A Novel Two-Pronged Attack on Huntington's
SKY-0515 is not just another shot on goal; it represents a sophisticated and targeted scientific approach. Developed using Skyhawk's proprietary SKYSTAR® platform, it is an orally-administered small molecule designed to modulate RNA, the messenger that carries instructions from DNA to the body's protein-making machinery. This allows it to target the very root of Huntington's disease with a unique dual mechanism.
First, the drug has shown a powerful ability to reduce the levels of mutant huntingtin protein (mHTT), the toxic protein that causes the disease. The data shows a dose-dependent reduction of mHTT in the blood of up to 62%. Professor Wild noted that this is "the greatest extent demonstrated by any therapeutic tested to date in patients."
Second, SKY-0515 also reduces the mRNA levels of a protein called PMS1 by 26%. PMS1 is a key driver of somatic CAG repeat expansion, a process that can accelerate disease progression in patients. By tackling both mHTT production and this expansion mechanism, SKY-0515 offers what Professor Wild calls a "potent combination for treating Huntington's disease via two of its core pathogenic mechanisms."
"Our goal for our Phase 1 study was to establish safety and biomarker activity," said Sergey Paushkin, Head of R&D at Skyhawk Therapeutics. "The continued strength of SKY-0515's biomarker response... underscores SKY-0515's potential as a best in class disease-modifying therapy for HD."
Navigating a Competitive and Challenging Landscape
The path to a Huntington's disease therapy is littered with discontinued programs and clinical trial failures, making Skyhawk's positive interim data all the more significant. The field includes several high-profile competitors pursuing different strategies. Roche is advancing Tominersen, an injected therapy that requires lumbar punctures, after a redesigned trial. PTC Therapeutics, in a major partnership with Novartis, is developing an oral drug, Votoplam, though recent data reportedly underwhelmed investors. Other approaches include gene therapies, which carry the promise of a one-time treatment but at a potentially multi-million dollar cost.
Against this backdrop, SKY-0515's profile appears highly competitive. Its status as a once-daily oral pill offers a profound quality-of-life advantage over infused or injected therapies, allowing patients to be treated at home. This convenience, combined with its unique dual-action mechanism and strong early data, positions Skyhawk as a serious contender in the high-stakes race for the first effective disease-modifying treatment.
From Lab to Global Trial: What's Next for SKY-0515
While the Phase 1 results are promising, they are still from an early-stage, open-label trial. The crucial next step is validation in a larger, more rigorous, placebo-controlled setting. Skyhawk is already well underway with this process. The company announced that its Phase 2/3 FALCON-HD trial has now expanded to sites worldwide, building upon its initial launch in Australia and New Zealand. More than 90 patients have already been dosed with SKY-0515 across its clinical programs, demonstrating significant momentum.
The FALCON-HD study will enroll hundreds of participants with early-stage Huntington's disease to definitively evaluate the safety and efficacy of SKY-0515 over a 12-month period. Success in this pivotal trial would pave the way for regulatory submission and potential approval.
For Skyhawk Therapeutics, SKY-0515 is the first drug from its RNA-modulating platform to reach this stage, serving as a critical validation of its underlying technology. The company has stated its intention to bring additional small molecule drugs for other rare neurological diseases into the clinic by the end of 2027, suggesting that today's news could be the first of many potential breakthroughs.
