Senti Bio Gets FDA Green Light for Pivotal AML Trial After 'Donor X' Boost
- 44% Overall Response Rate: SENTI-202 achieved a 44% overall response rate at the recommended Phase 2 dose, with all complete remissions being MRD-negative.
- 50% vs. 12.5% cCR Rate: Patients receiving 'Donor X' cells had a 50% composite complete remission (cCR) rate, compared to 12.5% for non-Donor X cells.
- <6 Months Median Survival: Patients with relapsed/refractory AML (R/R AML) face a median survival of less than six months.
Experts would likely conclude that Senti Bio's FDA approval for the pivotal AML trial, combined with the 'Donor X' breakthrough, significantly strengthens the therapeutic potential of SENTI-202, offering new hope for patients with limited treatment options.
Senti Bio Gets FDA Green Light for Pivotal AML Trial After 'Donor X' Boost
SOUTH SAN FRANCISCO, Calif. – May 14, 2026 – In a significant step forward for one of the most aggressive forms of blood cancer, Senti Biosciences announced today that it has received a clear path from the U.S. Food and Drug Administration (FDA) to advance its novel cell therapy, SENTI-202, into a pivotal trial for relapsed or refractory acute myeloid leukemia (R/R AML).
The announcement follows a successful high-level meeting with the agency and is bolstered by powerful new clinical data. An internal analysis revealed a specific donor characteristic, dubbed the 'Donor X' phenotype, that dramatically improves patient outcomes. This discovery has reshaped the company's manufacturing strategy and provides a major boost of confidence as it prepares for the final stages of clinical testing before a potential market approval.
For patients with R/R AML, who face a grim prognosis with a median survival of less than six months, the news represents a new wave of hope. Senti Bio now plans to launch a single-arm, multi-center registrational study, a streamlined clinical pathway often reserved for highly promising therapies in areas of extreme unmet medical need.
“This positive FDA RMAT meeting marks a transformational moment for Senti Bio and significantly advances our path toward potential registration of SENTI-202,” said Tim Lu, M.D., Ph.D., Chief Executive Officer and Co-Founder of Senti Bio. “We believe this milestone further validates both our Gene Circuit platform and the differentiated therapeutic potential of Logic Gated cell therapies.”
A Clearer Path for a Devastating Disease
The FDA's feedback came during a Type B Regenerative Medicine Advanced Therapy (RMAT) meeting. The RMAT designation is a high honor in biotech, granted by the FDA only to regenerative medicine therapies that show preliminary clinical evidence of addressing serious or life-threatening diseases. It provides a sponsor with intensive guidance and an expedited pathway, including the potential for rolling reviews and accelerated approval.
Senti Bio's ability to proceed with a single-arm trial is a direct result of this designation and the strength of its Phase 1 data. Unlike traditional randomized controlled trials that compare a new drug against a standard of care, a single-arm study enrolls a single group of patients who all receive the experimental therapy. This approach is typically accepted by regulators only when a disease is severe, existing treatments are largely ineffective, and the new therapy shows a profoundly strong signal of efficacy. This is precisely the case for R/R AML, where patients have exhausted most available options.
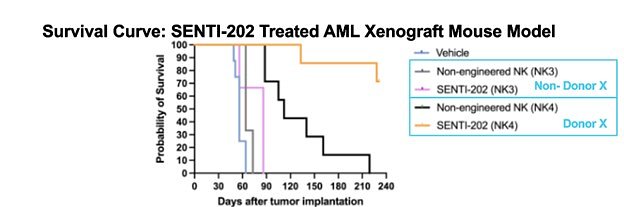
The durability of the responses seen so far has been a cornerstone of the program's success. The company reported that complete remissions are holding, with some patients remaining in remission for over 21 months—a remarkable duration for this disease. At the recommended Phase 2 dose, SENTI-202 achieved an overall response rate of 44%, with all complete remissions being MRD-negative, meaning no detectable cancer cells remained.
The 'Donor X' Breakthrough: Optimizing a Cellular Weapon
Perhaps the most compelling part of the update is the discovery of the 'Donor X' attribute. SENTI-202 is an allogeneic, or “off-the-shelf,” CAR-NK therapy, meaning it uses natural killer (NK) cells from healthy donors rather than a patient's own cells. In an exploratory analysis of its Phase 1 trial, Senti Bio’s scientists identified a specific characteristic in some donors that made their NK cells significantly more effective cancer killers.
The results were stark: patients who received SENTI-202 manufactured from 'Donor X' cells had a composite complete remission (cCR) rate of 50%. In contrast, patients who received cells from non-Donor X sources saw only a 12.5% cCR rate. This fourfold increase in deep responses provides a powerful tool for optimizing the therapy.
As a result of the finding, the company has made a decisive strategic pivot: all future manufacturing of SENTI-202, including the material for the upcoming pivotal trial, will be sourced exclusively from donors with the 'Donor X' phenotype. Senti Bio notes this attribute is present in approximately 50% of adult donors, ensuring a stable supply chain for the therapy.
While the specific biological markers of 'Donor X' are a closely guarded company secret, the strategy aligns with a major trend in cell therapy research. Scientists are increasingly focused on identifying and selecting for superior donor characteristics to enhance the potency and reliability of allogeneic treatments. This discovery is a powerful validation of Senti Bio's deep expertise in cell biology and manufacturing.
“The FDA feedback provides important clarity around our registrational development strategy and further supports our conviction in the SENTI-202 program,” said Kanya Rajangam, M.D., Ph.D., Chief Medical Officer of Senti Bio. “We are in a strong position as we prepare for our clinical trials with the identification of a donor phenotype that correlates with increased activity and continues to support SENTI-202’s allogeneic manufacturing.”
Precision Engineering in a Competitive Field
SENTI-202 is engineered with Senti Bio's proprietary Gene Circuit platform, which gives the therapy its “Logic Gated” capabilities. The CAR-NK cells are designed to recognize and kill cancer cells expressing either CD33 or FLT3, two common markers on AML cells. Critically, a built-in “NOT GATE” instructs the cells to spare healthy bone marrow cells that also express CD33, a design intended to reduce the severe side effects that have hampered other therapies targeting this marker. The therapy also includes a calibrated-release version of the cytokine IL-15 to help the engineered cells persist and expand in the body.
This sophisticated engineering, now combined with optimized donor selection, is how Senti Bio aims to distinguish itself in a competitive field. Other companies, like Fate Therapeutics and Nkarta Therapeutics, are also developing off-the-shelf NK cell therapies for AML. However, Senti Bio is betting that its multi-layered approach of precision targeting, built-in safety switches, and now, a highly potent donor source, will give SENTI-202 a decisive clinical advantage.
The de-risking effect of the FDA meeting and the 'Donor X' discovery provides significant momentum for the company as it moves toward the market. With a clear regulatory path and a refined manufacturing process, Senti Bio is poised to rapidly advance what could become a transformative treatment for patients who currently have very few places to turn.
📝 This article is still being updated
Are you a relevant expert who could contribute your opinion or insights to this article? We'd love to hear from you. We will give you full credit for your contribution.
Contribute Your Expertise →