New IBS Diet Shatters Pain Relief Benchmarks, Offers Palatable Hope
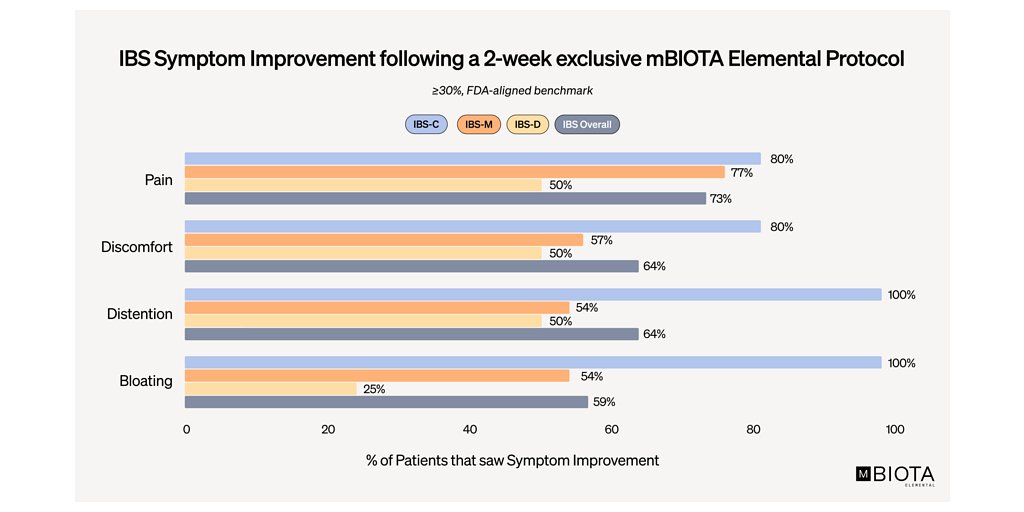
- 73% of IBS patients experienced at least a 30% reduction in daily abdominal pain after two weeks of using mBIOTA Elemental™.
- 50% of patients achieved a 50% or greater reduction in pain, surpassing FDA benchmarks.
- 100% patient compliance rate over a two-week exclusive regimen, a landmark achievement in elemental diet research.
Experts view mBIOTA Elemental™ as a groundbreaking advancement in IBS treatment, offering clinically meaningful pain relief and high patient adherence, with potential to become a first-line therapeutic option.
New IBS Diet Shatters Pain Relief Benchmarks, Offers Palatable Hope
CHICAGO, IL – May 04, 2026 – New research presented at the prestigious Digestive Disease Week (DDW) 2026 conference has unveiled stunningly effective results for a novel medical food designed for Irritable Bowel Syndrome (IBS), a condition that affects up to 15% of the global population. The findings on mBIOTA Elemental™, developed by Santa Monica-based mBIOTA Labs, demonstrate pain reduction levels that significantly surpass established FDA benchmarks, signaling a potential paradigm shift in how the notoriously difficult-to-manage gastrointestinal disorder is treated.
In a post-hoc analysis of a prospective trial, 73% of IBS patients using the elemental diet for two weeks experienced at least a 30% reduction in daily abdominal pain. More strikingly, half of the patients achieved a 50% or greater reduction in pain—a result that dramatically exceeds the 30% improvement benchmark the FDA considers clinically meaningful. These results, presented in a poster at DDW, suggest a powerful new tool for clinicians and a new horizon of hope for patients across all three IBS subtypes: constipation-dominant (IBS-C), diarrhea-dominant (IBS-D), and mixed (IBS-M).
The Palatability Breakthrough
For decades, elemental diets—nutritionally complete, pre-digested formulas that give the digestive system a rest—have been a powerful but dreaded therapeutic option. Their notoriously unpalatable taste has led to extremely low patient adherence, often making them a non-starter for all but the most severe cases. mBIOTA Labs appears to have solved this critical problem.
Using a proprietary Amino Taste Modification Technology (ATMT), the company developed a formula that patients can not only tolerate but adhere to. The proof lies in the data from the foundational prospective trial conducted at Cedars-Sinai, which focused on patients with Small Intestinal Bacterial Overgrowth (SIBO) and Intestinal Methanogen Overgrowth (IMO). In that study, mBIOTA Elemental™ achieved a 100% patient compliance rate over a two-week exclusive regimen. This perfect adherence is virtually unheard of in elemental diet research and is the key that unlocks the diet's therapeutic potential.
"The challenge with these diets has always been getting patients to actually consume them for the required duration," explained one gastroenterologist not affiliated with the study. "A 100% compliance rate is a landmark achievement. It suggests the product is not just theoretically effective but practically usable, which could make it a first-line consideration rather than a last resort."
Beyond Symptom Relief: Modulating the Microbiome
The new research presented at DDW delves deeper than just symptom relief. An oral presentation detailed how the two-week diet actively modulates the gut microbiome, the complex community of microorganisms living in the digestive tract. The analysis confirmed that the diet is a key driver of symptom relief by altering the gut environment, particularly in patients with microbial overgrowth syndromes like SIBO and IMO.
The study found that populations of microbial species associated with dysbiosis, including Methanobrevacter smithii (a key methanogen in IMO) and the fungus Candida albicans, were significantly decreased following the two-week diet. Perhaps more importantly, these troublesome populations remained suppressed even after patients reintroduced a regular diet, suggesting a lasting reset of the gut ecosystem rather than a temporary fix.
This mechanistic insight, combined with the broad symptom relief, paints a comprehensive picture. The diet appears to work by starving overgrown, problematic microbes while providing complete, easily absorbed nutrition to the patient. The benefits were widespread, with 82% of all IBS patients in the analysis achieving a statistically significant improvement in at least one key symptom, including pain, bloating, distention, or discomfort.
A Strategic Path Through the 'Medical Food' Category
Instead of pursuing the long and arduous path of pharmaceutical drug approval, mBIOTA Labs is positioning its elemental diet as a 'medical food.' This specific regulatory classification, defined by the FDA under the Orphan Drug Act, is for products intended for the dietary management of a disease under physician supervision. This pathway allows for a faster route to market than a prescription drug but requires that the product addresses distinctive nutritional requirements backed by scientific principles.
This strategy allows mBIOTA Elemental™ to be used by clinicians now, based on the compelling evidence of its efficacy and safety. It occupies a unique space between less-regulated dietary supplements and rigorously tested pharmaceuticals, offering a clinically validated, non-drug intervention for complex conditions. By focusing on a palatable, evidence-based nutritional solution, the company is tapping into a growing demand for therapies that work with the body's own systems.
"We are extremely proud to share the IBS patient symptom reduction findings at DDW 2026 as yet another rare achievement across GI clinical trials," said Nicola Wodlinger, CEO of mBIOTA Labs, in the company's press release. "This new clinical evidence validates mBIOTA Elemental Diet as an effective therapeutic tool for IBS."
The company has stated its commitment to further research, with the strong efficacy findings signaling a green light for future, larger-scale clinical trials specifically designed for IBS. Such trials could further solidify the diet's role and potentially redefine the therapeutic outcome expectations for a condition that has long frustrated both patients and their doctors. The results signal a promising future where nutritional science plays a central role in managing complex gastrointestinal disorders.
📝 This article is still being updated
Are you a relevant expert who could contribute your opinion or insights to this article? We'd love to hear from you. We will give you full credit for your contribution.
Contribute Your Expertise →