Neuronoff's Injectrode Aims to Revolutionize Neuromodulation
- $10 billion: The global neuromodulation market is projected to surpass this value by 2030.
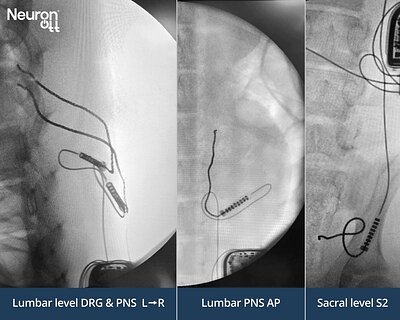
- 3 nerve targets: Neuronoff's Injectrode successfully stimulated the Lumbar Dorsal Root Ganglion, Lumbar Dorsal Ramus, and Sacral S2 foramen using a single IPG.
- 1.5mm needle: The Injectrode is delivered through a minimally invasive procedure using a needle of this diameter.
Experts would likely conclude that Neuronoff's Injectrode represents a transformative advancement in neuromodulation, offering a minimally invasive, versatile solution that expands the capabilities of existing IPG systems and could significantly broaden treatment options for chronic pain and other nerve-related conditions.
Neuronoff's Injectrode Aims to Revolutionize Neuromodulation
CLEVELAND, OH – March 16, 2026 – Clinical-stage company Neuronoff has announced a significant milestone that could fundamentally alter the landscape of chronic pain management and nerve-related therapies. The company revealed the successful completion of a chronic large animal study for its IPG-compatible Injectrode® platform, demonstrating that a single, standard implantable pulse generator (IPG) can selectively stimulate three distinct nerve targets through minimally invasive, needle-delivered electrodes.
This breakthrough challenges the decades-long paradigm of neuromodulation, which has been dominated by surgically intensive procedures primarily focused on the spinal cord. By proving the ability to connect its injectable electrodes to the same power sources used in mainstream spinal cord stimulation (SCS), Neuronoff is positioning its technology not as a replacement for existing systems, but as a massive expansion of their capabilities, potentially unlocking a multi-billion dollar market of peripheral nerve targets that have remained largely out of reach.
A New Era for Nerve Stimulation
For years, patients with chronic pain or other nerve-related conditions faced a stark choice: manage with pharmaceuticals or undergo major surgery to implant a stimulation device. Traditional neuromodulation systems, while effective for many, require surgeons to create pockets for generators and tunnel leads to specific sites on the spinal cord. Neuronoff’s Injectrode platform reimagines this process entirely.
The technology is delivered through a needle approximately 1.5mm in diameter—not much larger than those used for common injections. The electrode itself is a helical platinum-iridium wire that, once injected, anchors itself into the surrounding tissue. This inherent stability aims to address a persistent problem in the industry: lead migration, which is a leading cause of device failure and re-operation. The procedure requires no sutures and leaves no significant scarring, transforming the patient experience from a major surgical event to a minimally invasive, clinic-based procedure.
In the recent study, Neuronoff demonstrated this capability by placing tripolar Injectrodes at three separate, anatomically distinct sites routinely targeted by pain physicians: the Lumbar Dorsal Root Ganglion (DRG), the Lumbar Dorsal Ramus, and the Sacral S2 foramen. Using a single, remotely controlled IPG, the company proved it could independently and selectively stimulate each target. This multi-channel capability from a single power source is a critical step in showing the platform's versatility.
"Spinal cord stimulation (SCS) has been the primary market for IPGs for decades, but the vast majority of treatable nerve targets with easy access are peripheral," said Manfred Franke, CEO of Neuronoff, in a statement. "The IPG-Injectrode removes the surgical barrier in PNS... Any IPG can now reach any nerve a physician can access with a needle — and that is a fundamentally different addressable market."
Reshaping the Neuromodulation Market
The strategic genius of Neuronoff's approach lies in its integration with, rather than opposition to, the established market. The global neuromodulation market is a significant and growing space, with projections estimating its value will surpass $10 billion by 2030, driven by an aging population and the rising prevalence of neurological disorders. Major players like Medtronic, Abbott, and Boston Scientific have invested billions in developing sophisticated IPGs—the battery packs and processing units for these systems.
Neuronoff’s Injectrode connects to these existing IPGs using industry-standard Bal Seal connectors. This 'IPG-agnostic' design means that any manufacturer's device can, in theory, be used to power the Injectrode system. It effectively offers these industry giants a way to vastly expand the addressable market for their existing hardware without undertaking the massive R&D costs of developing a comparable injectable electrode system themselves. Instead of just powering spinal cord stimulators, their IPGs could soon be used to treat a wide array of peripheral nerve conditions, from chronic lower back pain originating in the dorsal ramus to sacral nerve issues.
This opens the door to a new ecosystem of treatment, where interventional pain physicians can use their needle-based skills to deploy advanced, implantable technology that was previously the exclusive domain of specialized surgeons.
The Patient Journey Reimagined
Perhaps the most compelling aspect of Neuronoff's strategy is its focus on a seamless, patient-centric continuum of care. The company is developing its platform along two parallel paths designed to meet patients where they are in their treatment journey.
The first step is an externally powered system, for which Neuronoff plans a 510(k) FDA submission in mid-2026. This system allows a patient to undergo a minimally invasive trial of the Injectrode, similar in complexity to a diagnostic nerve block. They can experience the therapeutic effects of neuromodulation without committing to a permanent implant. This 'try before you buy' approach dramatically lowers the barrier to entry for patients who may be hesitant about irreversible surgical procedures.
"Our external EPG system and IPG-compatible system are designed as a continuum," Franke explained. "The patient journey begins with a minimally invasive step not unlike a steroid injection or needle-based RF ablation — no suturing, no scarring, fully compatible with clinic workflows."
For patients who experience significant relief and wish to proceed with a long-term solution, the second stage comes into play: the IPG-compatible system. This allows for a straightforward conversion to a fully implanted, battery-powered chronic therapy. Because the platform is built on existing hardware standards, this transition is designed to be a natural and procedurally simple next step.
Paving a Clear Path to Market
Beyond the technological innovation, Neuronoff has demonstrated a shrewd understanding of the commercial realities of the medical device industry. A novel technology is only valuable if physicians can get paid to use it. To that end, the Injectrode platform has been designed from the ground up to align with established CPT reimbursement codes, specifically 64555 (for percutaneous implantation of a neurostimulator electrode) and 64590 (for insertion of a neurostimulator pulse generator).
By leveraging this existing reimbursement infrastructure, Neuronoff bypasses one of the most significant and time-consuming hurdles that new medical technologies face. Clinicians will not need to fight with payers over unlisted procedure codes, which should dramatically accelerate adoption upon regulatory approval. The company's mid-2026 submission target for its external system appears feasible, with FDA 510(k) review times for similar devices averaging around six months. This could put the initial version of the Injectrode on the market by early 2027.
This regulatory path is further bolstered by a successful first-in-human trial completed in January 2024, which demonstrated the safety and functionality of the externally powered system in patients with chronic lower back pain.
Beyond Pain: The Future of Injectable Neuromodulation
While chronic pain is the immediate and largest target market, Neuronoff's vision extends far beyond it. The press release and company materials allude to a future where the Injectrode platform could address a host of other conditions, including those related to autonomic, bladder, cardiac, and inflammatory systems.
Sacral nerve stimulation is already an established therapy for overactive bladder, and the Injectrode’s ability to target these nerves with a minimally invasive approach could expand patient access. Similarly, the emerging field of cardiovascular neuromodulation, which explores electrical stimulation for conditions like heart failure and refractory angina, could be a fertile ground for this technology. By providing precise, targeted stimulation to nearly any peripheral nerve accessible by a needle, the platform has the potential to become a foundational tool for a new generation of bioelectronic medicines, offering hope for a wide range of chronic conditions that are currently difficult to manage.
