Hope for ALS: Neurizon Begins Pivotal Trial for Promising Drug NUZ-001
- 31% slower rate of functional decline in ALS patients treated with NUZ-001 compared to historical controls in a Phase 1 study.
- Nearly 50% slower decline in respiratory function observed with NUZ-001 treatment.
- 76% reduction in risk of death associated with NUZ-001 in preliminary data.
Experts view the initiation of the HEALEY ALS Platform Trial for NUZ-001 as a significant step forward in ALS research, offering cautious optimism based on promising early data while acknowledging the high failure rate in the field.
Hope for ALS: Neurizon Begins Pivotal Trial for Promising Drug NUZ-001
MELBOURNE, Australia – February 26, 2026 – In a significant development for the amyotrophic lateral sclerosis (ALS) community, Australian biotechnology company Neurizon Therapeutics has announced the dosing of the first participant in a pivotal U.S. trial for its lead drug candidate, NUZ-001. The study is part of the groundbreaking HEALEY ALS Platform Trial, an innovative research model designed to accelerate the search for effective treatments for the devastating neurodegenerative disease.
The initiation of this adaptive Phase 2/3 clinical trial marks a critical milestone for both Neurizon and the thousands of individuals living with ALS, a relentlessly progressive condition with limited therapeutic options. NUZ-001 now joins a select group of promising compounds being evaluated under the prestigious HEALEY framework, offering a new beacon of hope in a field defined by urgent unmet medical need.
A Repurposed Drug with a Novel Approach
At the heart of this new trial is NUZ-001, an oral small molecule with an unconventional backstory. Originally developed as a veterinary deworming agent known as monepantel, the drug has been repurposed for its potential neuroprotective properties. Its proposed mechanism of action targets a fundamental cellular process implicated in the progression of neurodegenerative diseases like ALS.
NUZ-001 is an inhibitor of the mTOR signaling pathway. In healthy cells, mTOR helps regulate growth and metabolism. However, in conditions like ALS, its overactivity can suppress autophagy—the cell's essential "recycling" system responsible for clearing out damaged proteins and other cellular debris. The toxic accumulation of these proteins is a key pathological hallmark of ALS. By inhibiting mTOR, NUZ-001 is hypothesized to enhance autophagy, helping neurons clear these harmful aggregates and potentially slowing the disease's destructive course.
This scientific rationale is supported by encouraging preliminary data from a small Phase 1 study (the MEND study) involving 12 ALS patients in Australia. Over a 12-month period, participants treated with NUZ-001 showed a 31% slower rate of functional decline compared to historical controls. The treatment also appeared to slow the decline in respiratory function by nearly 50% and was associated with a significant survival benefit, reducing the risk of death by over 76%. Throughout the study and a subsequent open-label extension, NUZ-001 was found to be safe and well-tolerated. These promising, albeit early, signals provided the strong scientific merit required for its selection into the highly competitive HEALEY trial.
The HEALEY Platform: Revolutionizing ALS Research
Neurizon’s NUZ-001 is not just entering any clinical trial; it is joining a revolutionary research ecosystem. The HEALEY ALS Platform Trial, led by the Sean M. Healey & AMG Center for ALS at Mass General Brigham, represents a paradigm shift in how potential therapies are tested. Instead of the traditional, slow, and costly model of running separate, single-drug trials, the platform evaluates multiple investigational drugs simultaneously under a single master protocol.
This innovative design shares infrastructure, resources, and, most importantly, a common placebo group across more than 70 top clinical trial sites in the United States. This structure dramatically increases efficiency, reducing trial timelines by as much as 50% and costs by 30%. For patients, it increases the probability of receiving an active investigational drug rather than a placebo and broadens access to cutting-edge research.
"We look forward to working with Neurizon on this new regimen in the HEALEY ALS Platform Trial," commented Professor Merit Cudkowicz, the trial's Principal Investigator and Director of the Sean M. Healey & AMG Center for ALS. "Beginning enrolment is a significant step for the regimen, and would not be possible without the dedication of people living with ALS and their families, collaborators, and our top trials sites."
The platform's rigorous design has established it as a critical proving ground. While it accelerates development, it does not lower the scientific bar. Several drug candidates have been tested on the platform, with some failing to meet their endpoints, thereby demonstrating the trial's ability to efficiently and decisively identify which therapies warrant further investment.
High Stakes in a Challenging Landscape
The initiation of this registrational trial represents a high-stakes moment for Neurizon Therapeutics. A successful outcome could provide the clinical evidence needed to seek regulatory approval from the U.S. Food and Drug Administration (FDA), potentially transforming the small Australian biotech into a major player in the neurology space.
"The dosing of the first participant in Regimen I of the HEALEY ALS Platform Trial marks a defining milestone for Neurizon and for NUZ-001," said Dr. Michael Thurn, the company's Managing Director and Chief Executive Officer. "This study represents our registrational trial in ALS - a rigorous, adaptive Phase 2/3 program designed to generate the clinical evidence required to support potential regulatory submissions."
The urgency of this work is felt most acutely by the ALS community. The recent voluntary market withdrawal of Relyvrio, a drug that failed to show efficacy in a larger Phase 3 trial after receiving an earlier approval, serves as a stark reminder of the immense challenges in developing effective ALS treatments. The current standard-of-care drugs, such as Riluzole and Edaravone, offer only modest benefits in slowing disease progression and do not halt the disease.
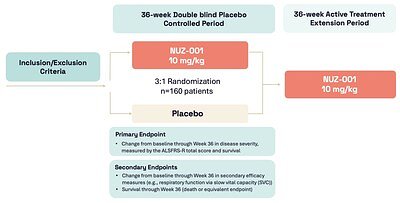
For the approximately 160 participants who will enroll in the NUZ-001 arm, known as Regimen I, the trial involves a 36-week, double-blind treatment period. Participants will be randomized on a 3-to-1 basis to receive either daily NUZ-001 or a placebo. This design, coupled with an active treatment extension phase, ensures that the trial gathers robust data on efficacy and safety while maximizing participants' access to the potential therapy.
Dr. Thurn expressed deep gratitude for the trial's participants. "For people living with ALS, urgency matters. Through this pivotal study, we are advancing NUZ-001 with scientific discipline, operational focus, and deep respect for the ALS community as we work toward delivering meaningful progress in this devastating disease." With enrollment expected to be completed in the second half of 2026, patients, physicians, and investors will be watching closely as this promising candidate is put to the ultimate test.
