Gore's New TIPS Device Gets CE Mark, Heralding Precision Liver Care
- Expanded Diameter Range: The GORE® VIATORR® TIPS Endoprosthesis now offers an adjustable diameter range of 6–10 mm, up from the previous 8–10 mm.
- Class III CE Mark Approval: The device has achieved the highest risk category certification under the EU’s Medical Device Regulation (MDR 2017/745).
- 500+ Publications: The VIATORR® Endoprosthesis is supported by over 500 clinical publications, underscoring its reliability.
Experts agree that the expanded diameter range of the GORE® VIATORR® TIPS Endoprosthesis represents a significant advancement in portal hypertension treatment, enabling more precise and personalized care to reduce complications like hepatic encephalopathy and cardiac overload.
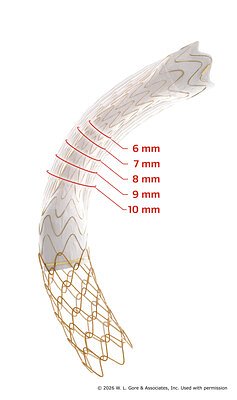
Gore's New TIPS Device Gets CE Mark, Heralding Precision Liver Care
PUTZBRUNN, Germany – January 22, 2026 – W. L. Gore & Associates has secured a pivotal CE mark for an expanded diameter range of its GORE® VIATORR® TIPS Endoprosthesis with Controlled Expansion, a move poised to significantly refine the treatment of portal hypertension. The approval expands the device's adjustable diameter from its previous 8–10 mm range to a more versatile 6–10 mm, offering interventional radiologists unprecedented control to customize a critical procedure for patients with severe liver disease.
This regulatory milestone allows for a more individualized approach to creating a transjugular intrahepatic portosystemic shunt (TIPS), a channel within the liver that reduces dangerously high blood pressure in the portal vein system. By enabling physicians to select a smaller initial shunt diameter, the updated device directly addresses one of the most persistent challenges in portal hypertension management: balancing effective pressure reduction with the risk of severe, life-altering complications.
A New Era of Precision in Portal Hypertension Treatment
The TIPS procedure is a lifeline for patients suffering from complications of advanced liver disease, such as variceal bleeding and refractory ascites. However, its success hinges on a delicate balance. A shunt that is too large can divert an excessive amount of blood from the portal vein, leading to complications like hepatic encephalopathy—a serious neurological condition causing confusion and cognitive decline—and cardiac overload. Conversely, a shunt that is too small may fail to adequately relieve portal pressure, leaving the patient vulnerable to the very complications the procedure was meant to prevent.
This clinical dilemma has long forced physicians to walk a fine line. The introduction of the 6-10 mm GORE® VIATORR® device provides a powerful new tool to navigate this challenge. The ability to create a smaller, more precise shunt allows clinicians to tailor the intervention to the specific anatomical and physiological needs of each patient.
“My research and clinical experience... have consistently shown that optimizing shunt diameter is critical for improving outcomes in portal hypertension,” stated Professor Filippo Schepis, Head of the Hepatic Hemodynamic Lab at the University Hospital of Modena, Italy. He emphasized the clinical significance of this advancement, noting, “Smaller-diameter devices may significantly reduce complications such as hepatic encephalopathy and cardiac overload. The VIATORR® Device with controlled expansion technology represents an important evolution, enabling precise tailoring of shunt size to individual patient needs. This is not just a technical refinement – it could provide a meaningful improvement for those living with portal hypertension.”
Navigating a Strict Regulatory Landscape
Achieving CE mark approval under the European Union’s stringent Medical Device Regulation (MDR 2017/745) is a significant validation of a device's safety and performance. The GORE® VIATORR® TIPS Endoprosthesis is classified as a Class III device, the highest risk category, which subjects it to the most rigorous pre-market scrutiny by a Notified Body. This certification confirms that the device has met comprehensive standards for quality, safety, and clinical efficacy, providing assurance to both clinicians and patients across the European Economic Area.
The device's approval is built on a long legacy of clinical trust. The VIATORR® Endoprosthesis has been used in clinical practice for over two decades, supported by a body of evidence that includes more than 500 publications. This extensive history underscores its reliability and durable performance. A key feature contributing to this longevity is its construction with Gore’s proprietary expanded polytetrafluoroethylene (ePTFE) graft lining. This material is specifically engineered to resist permeation by bile and mucin, which are common culprits in the loss of shunt patency over time, often leading to the need for re-intervention.
Solidifying Market Position and Healthcare Impact
With this CE mark, Gore strengthens its leadership position in a competitive medical device market that includes major players like Cook Medical and Medtronic. The unique combination of controlled expansion technology and a broader diameter range in a single device offers a distinct advantage. As noted by Roberto Miraglia of ISMETT in Palermo, Italy, this innovation “is expected to represent a significant advancement in the management of patients with complications of portal hypertension.”
Beyond the clinical benefits, the new device carries substantial implications for healthcare economics. By potentially reducing the incidence of severe complications, it can lead to significant cost savings for health systems. Fewer complications translate to shorter hospital stays, a lower likelihood of costly re-interventions, and a reduced burden on long-term care resources. This aligns with the growing global trend towards value-based healthcare, where the focus is on achieving better patient outcomes more efficiently.
The logistical benefits for hospitals are also notable. “Now, with a broader diameter range in one single device, we're not only improving control during implantation — we also deliver more options to physicians without increasing inventory,” said Thomas Wiersma, an EMEA Product Specialist at Gore. This streamlined approach simplifies hospital procurement and inventory management while expanding clinical capabilities.
A New Horizon for Patient Quality of Life
For the millions of people worldwide living with advanced liver disease, the impact of this technological refinement is deeply personal. Complications like hepatic encephalopathy can be devastating, robbing individuals of their cognitive function, independence, and quality of life. The prospect of a treatment that can effectively manage portal hypertension while minimizing such debilitating side effects offers new hope.
By enabling a more personalized and less aggressive shunting strategy when appropriate, the expanded VIATORR® device has the potential to dramatically improve the patient experience. Patients may face a lower risk of cognitive decline and cardiac strain, allowing them to maintain a higher level of daily function and overall well-being. This patient-centric approach transforms a technical innovation into a tangible improvement in human lives.
Gore's commitment to validating these benefits is further demonstrated by an ongoing post-market clinical study (NCT05661331). The trial is designed to confirm the device's performance and safety over a three-year period in a real-world setting, with a specific focus on collecting data on patients' quality of life following the procedure. The results of this study, expected to complete in the coming years, will provide further evidence of the long-term impact of this tailored approach to treating portal hypertension.
