French Bioprinter Bets $27M on Philly to Fix Flawed Drug Trials
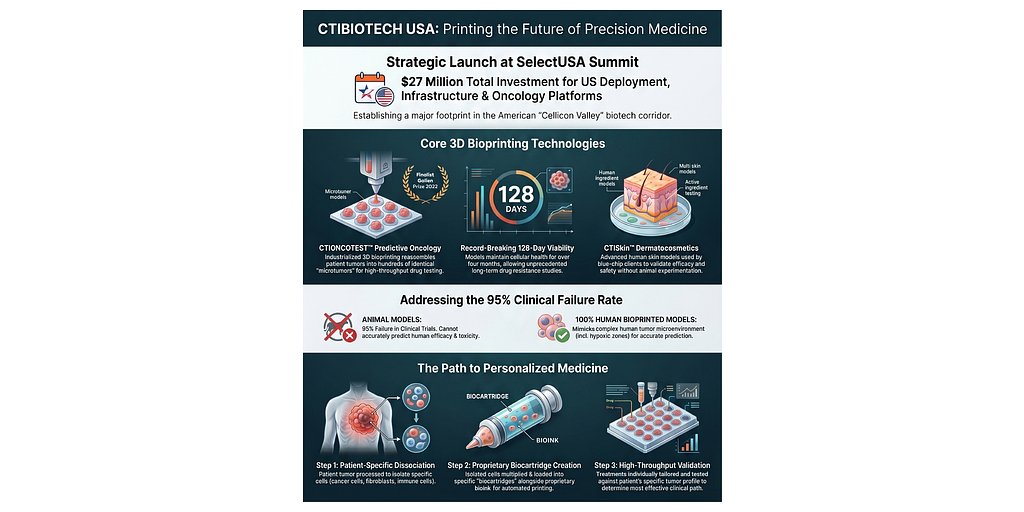
- $27M Investment: CTIBIOTECH™ is investing $27 million to establish its North American headquarters in Philadelphia, including $12 million in operational funding and $15 million in intellectual property assets.
- 95% Failure Rate: Currently, 95% of new cancer drugs fail during human clinical trials, costing the industry billions.
- $19B in Capital: Philadelphia has attracted nearly $19 billion in life sciences capital, solidifying its status as a global epicenter for cell and gene therapy.
Experts agree that CTIBIOTECH’s 3D bioprinting technology represents a significant advancement in drug development, offering more accurate human tissue models that could drastically reduce the high failure rate of clinical trials and accelerate the delivery of effective therapies.
French Bioprinter Bets $27M on Philly to Fix Flawed Drug Trials
PHILADELPHIA, PA – May 04, 2026 – In a significant move poised to reshape the landscape of American drug development, French bioprinting pioneer CTIBIOTECH™ has announced a $27 million investment to establish its North American headquarters in Philadelphia. The launch of CTIBIOTECH USA™, unveiled at the prestigious SelectUSA Investment Summit in Washington, D.C., marks a major transatlantic expansion aimed squarely at solving one of modern medicine’s most expensive problems: the staggering failure rate of new drugs.
Operating from the heart of Philadelphia's burgeoning life sciences corridor, aptly nicknamed 'Cellicon Valley,' the new subsidiary will leverage its cutting-edge 3D bioprinting technology to produce advanced human tissue models for the pharmaceutical, biomedical, and dermatocosmetics industries. By providing a local source for these highly sophisticated testing platforms, the company intends to accelerate research and development, reduce reliance on animal testing, and ultimately improve the success rate of clinical trials for life-saving therapies.
The investment is substantial, comprising approximately $12 million in direct operational funding over the next five years and an additional $15 million in intellectual property assets being deployed in the U.S. market. This strategic entry underscores a growing confidence in both the technology's potential and Philadelphia's capacity to foster groundbreaking biomedical innovation.
'Cellicon Valley' Welcomes a New Player
The decision to anchor its U.S. operations in Philadelphia is no coincidence. The region has solidified its reputation as a global epicenter for cell and gene therapy, consistently ranking among the top life science markets in the nation. Recognized by the Economic Development Administration as a tech hub for precision medicine, the area is home to a dense ecosystem of world-class academic institutions, a deep talent pool, and a robust network of established and emerging biotech firms.
With over 70,000 life sciences employees and a recent history of attracting nearly $19 billion in capital, Philadelphia provides fertile ground for a company like CTIBIOTECH. The new U.S. subsidiary will operate in association with BioLabs for Advanced Therapeutics, a state-of-the-art incubator space designed to support biotech innovation. This proximity to other leaders in the field, including Thermo Fisher Scientific's collaboration center within the same facility, creates a powerful synergy for research and commercialization.
The expansion is the result of extensive collaboration between international partners. The press release acknowledged instrumental support from the U.S. Federal Government's SelectUSA program, the State of Pennsylvania, and the City of Philadelphia, all of which played a role in attracting the foreign direct investment. This concerted effort highlights a strategic push to cement the region's status as a premier destination for global life science leaders.
The Science of Printing Human Tissue
Founded in Lyon, France, in 2009, CTIBIOTECH has built its reputation on a visionary approach to creating functional human tissues for research. The company specializes in upcycling human surgical tissues—materials that would otherwise be discarded—and using them as a biological foundation for 3D bioprinting. This process allows them to construct complex, three-dimensional biological structures that more accurately mimic the microenvironment of tissues and organs within the human body.
This technology represents a monumental leap beyond traditional two-dimensional cell cultures, which are grown in flat layers in a petri dish and often fail to replicate the intricate cell-to-cell interactions that govern disease and drug responses in humans. CTIBIOTECH’s proprietary microtumor and human skin models, for example, provide researchers with highly predictive biological assays to test the efficacy and toxicity of new compounds.
“Currently, 95% of new cancer drugs fail during human clinical trials, costing the industry billions and delaying life-saving treatments,” stated Professor Colin McGuckin, President and Chief Scientific Officer of CTIBIOTECH, in the official announcement. “By bringing our revolutionary 3D bioprinting technologies... directly to North America, we are providing researchers with the highly predictive, human-derived biological assays they need to reliably test new therapies and advance personalized medicine.”
A New Front in the War on Disease
The implications of this technology are profound, particularly in oncology, where the drug development pipeline is notoriously leaky. The industry-wide 95% failure rate for cancer drugs has remained stubbornly high for decades, largely because preclinical models—including animal studies and 2D cell cultures—are poor predictors of human response. Drugs that appear promising in the lab often prove to be ineffective or toxic when they reach human trials.
By providing models that are biologically human and structurally three-dimensional, 3D bioprinting offers a way to “fail faster and cheaper.” Pharmaceutical companies can identify non-viable drug candidates earlier in the development process, allowing them to redirect resources toward more promising therapies. This not only saves immense time and money but also accelerates the journey of effective treatments from the lab to the patient.
Furthermore, the technology is a cornerstone of personalized medicine. In the future, it will be possible to create patient-specific tissue models using an individual’s own cells. A bioprinted microtumor derived from a patient's biopsy could be used to test a panel of chemotherapy drugs, identifying the most effective treatment for that person's unique cancer before the first dose is ever administered.
Dr. Nico Forraz, the company’s CEO, emphasized the strategic advantage of a U.S. presence. “By establishing our proprietary manufacturing and contract research capabilities here in the U.S., we are eliminating the geographic challenges of international research and delivering immediate, localized innovation to the North American pharmaceutical ecosystem,” he noted.
An Ethical Shift and Regulatory Tailwinds
Beyond its scientific and commercial advantages, CTIBIOTECH’s expansion aligns with a powerful ethical and regulatory movement away from animal testing. For decades, drug safety has been evaluated in animals, a practice that is not only costly and fraught with ethical concerns but also scientifically questionable due to physiological differences between species.
The U.S. regulatory environment is now actively encouraging a transition to what are known as New Approach Methods (NAMs), a category that includes 3D bioprinted tissues and organ-on-a-chip technologies. A pivotal moment came with the passage of the FDA Modernization Act 2.0, part of the Food and Drug Omnibus Reform Act of 2022, which officially removed the mandate for animal testing, allowing drug developers to use qualified alternative methods to establish a drug’s safety and effectiveness.
Following this legislative change, the FDA has been developing a clearer framework for the validation and acceptance of NAMs. This regulatory tailwind creates a receptive market for CTIBIOTECH’s offerings, providing pharmaceutical companies with both the incentive and the regulatory pathway to adopt these more humane and human-relevant testing platforms. This convergence of advanced technology, significant investment, and a modernizing regulatory landscape positions the company's U.S. launch not just as a corporate expansion, but as a critical development in the future of American healthcare innovation.
📝 This article is still being updated
Are you a relevant expert who could contribute your opinion or insights to this article? We'd love to hear from you. We will give you full credit for your contribution.
Contribute Your Expertise →