Endospan's NEXUS Stent Wins FDA Nod for High-Risk Aortic Repair
- 120,000+ people in the U.S. and Europe diagnosed annually with thoracic aortic arch disease
- 0.0% lesion-related mortality rate and 1.1% disabling stroke rate in the TRIOMPHE study
- $600 million estimated annual global market opportunity for this indication
Experts agree that the FDA approval of Endospan's NEXUS Stent Graft System represents a significant advancement in treating high-risk aortic arch disease, offering a minimally invasive alternative with strong clinical outcomes where traditional surgery was often too risky.
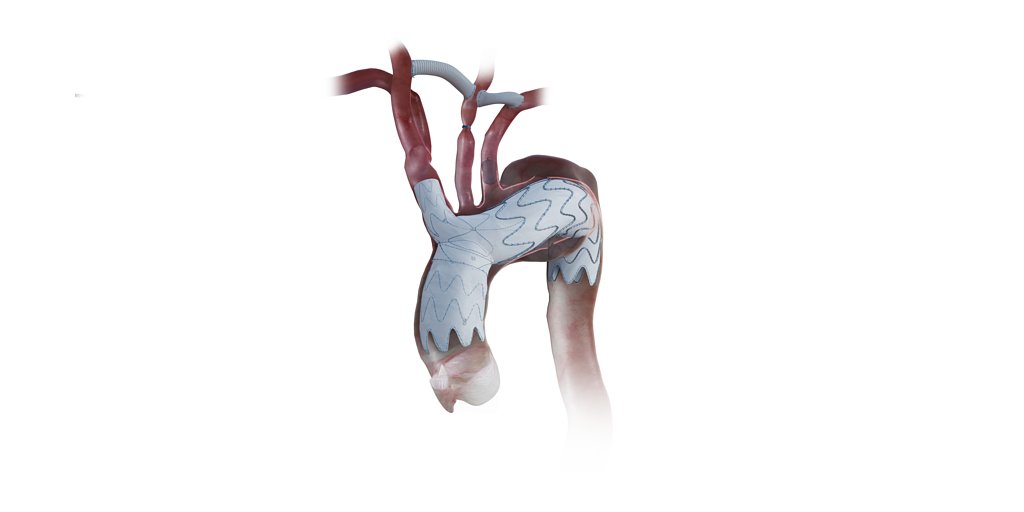
Endospan's NEXUS Stent Wins FDA Approval for Aortic Arch Repair
TEL AVIV, Israel – April 07, 2026 – The U.S. Food and Drug Administration (FDA) has granted approval to Endospan for its NEXUS® Aortic Arch Stent Graft System, a landmark decision that provides a new, minimally invasive treatment for patients with life-threatening aortic disease who are too frail for open-heart surgery. The approval paves the way for a U.S. commercial launch, offering a critical alternative for a vulnerable population that has long had limited options.
The NEXUS system is designed to repair complex conditions like aneurysms and chronic dissections in the aortic arch, the crucial, curved section of the body's main artery that supplies blood to the brain and upper body. This approval was based on compelling one-year results from the TRIOMPHE clinical study, which demonstrated the device's safety and effectiveness in high-risk patients.
Addressing a Critical Unmet Need in Aortic Care
Each year, more than 120,000 people in the United States and Europe are diagnosed with thoracic aortic arch disease. This includes dangerous conditions where the aortic wall weakens and bulges (aneurysm) or tears (dissection), creating a risk of catastrophic rupture. Yet, industry data reveals that only about a quarter of these individuals receive treatment.
The long-standing gold standard, open surgical repair, is a formidable procedure. It requires a large chest incision, stopping the heart, and rerouting blood flow through a heart-lung machine, often while cooling the body to protect the brain. For many patients, particularly the elderly or those with other significant health issues, the risks of mortality, stroke, and other major complications are simply too high. This has left a significant treatment gap, where thousands of patients are deemed inoperable and are managed with medication and monitoring, living under the constant threat of a fatal aortic event.
Endovascular repair, which involves threading a stent graft through blood vessels to reline and reinforce the aorta from within, has revolutionized treatment for other parts of the aorta. However, the unique, curved anatomy of the aortic arch and the critical arteries branching from it have made it the final frontier for this minimally invasive approach. The NEXUS system is specifically engineered to overcome these challenges, providing an "off-the-shelf" solution that does not require custom manufacturing for each patient.
The Science of Success: Inside the TRIOMPHE Study
The FDA's decision was anchored by robust clinical evidence from the TRIOMPHE study, a prospective, multicenter trial that evaluated NEXUS in 94 high-risk surgical patients. The study specifically targeted pathologies in Zone 0, the most challenging segment of the arch, which includes the ascending aorta just as it leaves the heart.
One-year results from the study were particularly impressive, showcasing the device's ability to provide a durable repair with a strong safety profile. The data revealed a 0.0% lesion-related mortality rate and a disabling stroke rate of just 1.1%—remarkably low figures for this complex patient group. Furthermore, core laboratory analysis confirmed excellent sealing of the device, with no instances of Type III or IV endoleaks, which are critical device-related failures.
"The anatomical design of the NEXUS System addresses many of the complexities that occur when treating the ascending and aortic arch,” said Dr. Brad Leshnower, a national co-principal investigator for the study. “I am pleased to now be able to offer this minimally invasive solution to patients with life threatening diseases involving the ascending aorta and arch who are at high risk for conventional open repair.”
The system's design is key to its success. NEXUS is a bimodular system with a low-profile 20F delivery catheter, one of the smallest available for such a complex procedure. Its catheter is pre-shaped to match the arch's curve, allowing surgeons to navigate the delicate anatomy with a single pass, minimizing manipulation and reducing the risk of dislodging plaque that could lead to a stroke. An integrated branch is also built into the stent graft to ensure continued blood flow to the brain.
Dr. Ross Milner, the study's other national co-principal investigator, praised the collaborative effort behind the trial. "On behalf of my co-Principal National Investigator Dr. Leshnower and me, I would like to thank the entire TRIOMPHE investigational body for their commitment, time and effort to make this technology available and expand our treatment options for delivering aortic care to the community," he stated.
Reshaping the Competitive Landscape
With this approval, Endospan enters a competitive but rapidly evolving market for endovascular aortic repair, estimated to have a global addressable opportunity of $600 million annually for this specific indication. The Israeli-based company now stands alongside major medical device players, but with a product that fills a distinct niche.
While competitors like W. L. Gore & Associates offer their own branched devices for the aortic arch, such as the GORE® TAG® Thoracic Branch Endoprosthesis (TBE), Endospan's NEXUS is distinguished by its focus on the entire arch, including the notoriously difficult-to-treat ascending aorta. Its "off-the-shelf" availability is another significant advantage, streamlining procedure planning compared to devices that may require customization and lead times. The positive results from treating chronic dissections in the TRIOMPHE trial further underscore its unique capabilities in managing some of the most complex aortic pathologies.
This FDA clearance, which follows a CE Mark approval in Europe, solidifies Endospan’s position as a key innovator. The company’s ability to successfully navigate the rigorous FDA approval process with strong clinical data signals its potential to capture a significant share of the market by serving patients who previously had no viable minimally invasive options.
A New Path to Market and a Lifeline for Patients
The journey from regulatory approval to widespread clinical use is the next critical step for Endospan. The company's path to the U.S. market is uniquely structured, with medical technology firm Artivion holding an option to acquire Endospan, exercisable within 90 days of the FDA approval. Artivion, a leader in cardiac and vascular surgery solutions, had previously provided financing to support the TRIOMPHE trial, indicating a long-standing strategic interest. An acquisition would provide Endospan with Artivion’s established sales force and distribution network, significantly accelerating the rollout of NEXUS to hospitals across the country.
This strategic alignment promises to quickly translate a regulatory milestone into tangible patient benefits. For physicians, it means having a new, reliable tool to treat what was once considered an insurmountable clinical challenge. For patients and their families, it represents a new horizon of hope.
“This is an exciting milestone for our team, and we are grateful for the dedication and partnership of all the physicians and clinical staff involved in the TRIOMPHE Study,” said Kevin Mayberry, CEO of Endospan. “NEXUS was designed specifically for the ascending and aortic arch... we are pleased, with the FDA approval, to be able to offer NEXUS to U.S. patients.”
The availability of a less invasive, off-the-shelf solution is poised to not only improve outcomes for high-risk individuals but also potentially expand the number of patients who can be treated, fundamentally changing the standard of care for complex aortic arch disease.
