Endometriosis Breakthrough: New Scan Promises to End Diagnostic Limbo
- 190 million women worldwide are affected by endometriosis, yet diagnosis often takes 8-12 years.
- The new scan correctly identified endometriosis in 14 out of 17 surgically confirmed cases, with no false positives.
- 80% of endometriosis cases involve superficial peritoneal endometriosis (SPE), which is often undetectable by standard scans.
Experts view this breakthrough as a potential paradigm shift in endometriosis care, offering a highly promising, non-invasive diagnostic tool that could significantly reduce diagnostic delays and improve patient outcomes.
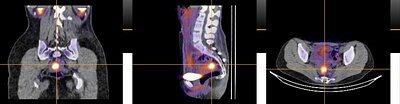
Endometriosis Breakthrough: New Scan Promises to End Years of Diagnostic Limbo
LONDON, UK – April 30, 2026 – For millions of women worldwide, the path to an endometriosis diagnosis is a long and painful journey, often spanning a decade of dismissed symptoms and invasive procedures. A groundbreaking new imaging technique, however, may finally offer a beacon of hope. Researchers from the University of Oxford and Serac Healthcare have announced promising results from a study on a novel radiotracer, 99mTc-maraciclatide, which has demonstrated the ability to non-invasively detect the disease with remarkable accuracy, potentially slashing diagnostic delays and transforming patient care.
The Agonizing Wait for an Answer
Endometriosis is a debilitating inflammatory disease where tissue similar to the uterine lining grows outside the uterus, causing chronic pain, inflammation, and potential infertility. The condition affects an estimated 190 million women of reproductive age globally—roughly one in ten. Yet, despite its prevalence, the journey to a formal diagnosis is notoriously fraught with delays, averaging between eight and twelve years.
Patients often report their debilitating pain being dismissed as "normal period pain," leading to years of suffering and a significant toll on mental health and quality of life. The current "gold standard" for a definitive diagnosis is a laparoscopic surgery, an invasive procedure that requires general anesthesia and carries inherent risks.
Conventional imaging methods like transvaginal ultrasound and MRI scans have proven unreliable for detecting many forms of the disease, particularly superficial peritoneal endometriosis (SPE). SPE is the earliest and most common form, found in approximately 80 percent of all surgically diagnosed cases, yet it often remains invisible to standard scans. The recently published study underscores this challenge: of the participants with surgically confirmed SPE, none had their disease detected by prior ultrasounds or MRIs performed within the last year. This diagnostic gap leaves countless women in a painful limbo, without answers or a clear treatment path.
A New Beacon of Hope: How the Scan Works
The new findings, published in the prestigious journal The Lancet Obstetrics, Gynaecology & Women's Health, detail the results of the Phase 2 DETECT study. The study evaluated an innovative molecular imaging agent, 99mTc-maraciclatide, which functions as a highly specific radiotracer.
The science behind the scan is elegant in its approach. The agent is designed to bind to a specific protein called αvβ3 integrin. This protein is heavily involved in angiogenesis—the formation of new blood vessels—a process that is a cardinal feature of the inflammation and growth of endometriosis lesions. When injected, the radiotracer travels through the body and accumulates at sites of active endometriosis, effectively "lighting them up" on a SPECT-CT scan.
The results of the DETECT study were striking. The imaging showed a high correlation with surgical findings across all types of endometriosis, including the hard-to-detect SPE lesions. In 19 patients who underwent both the scan and surgery, the imaging results correctly matched the surgical findings in 16 cases. The scan successfully identified endometriosis in 14 of the 17 surgically confirmed patients, including two rare cases of thoracic endometriosis in the chest cavity. Critically, the study reported no false positives, suggesting a high degree of specificity. Patients also found the procedure to be well-tolerated.
Voices from the Forefront of Research
The research team has hailed the results as a potential paradigm shift in endometriosis care. Dr. Tatjana Gibbons, the paper's lead author from the University of Oxford's Nuffield Department of Women's and Reproductive Health, highlighted its specific advantages. "These exciting findings indicate that maraciclatide offers a highly promising diagnostic and monitoring tool, particularly for superficial peritoneal endometriosis, which is the most common and yet the hardest type of endometriosis to identify," she stated.
This sentiment was echoed by senior investigators on the study. Professor Christian Becker, Co-Director of the Endometriosis CaRe Centre in Oxford, emphasized the urgent need for such an innovation. "Novel, non-invasive diagnostic tests for endometriosis are a global research priority," he said. "If these Phase II results are reproduced in the Phase 3 studies, maraciclatide has the potential to be an extremely valuable tool, as it could both reduce diagnostic delays and provide a validated endpoint for the development of new therapeutics."
Professor Krina Zondervan, another co-lead on the study, pointed to the transformative potential for both patients and researchers. "If these results are confirmed in larger Phase 3 studies, imaging with maraciclatide could transform clinical research and practice and potentially empower the development of treatments for women across the globe," she commented.
For Serac Healthcare, the company developing the agent, the publication marks a significant step forward. "The completion and publication of this clinical study is a major milestone," said David Hail, CEO of Serac Healthcare. "With FDA Fast Track Designation and agreed Phase 3 study designs, we are now advancing to validate these findings in larger trials and progress to regulatory submission."
The Path Forward: From Lab to Clinic
While the Phase 2 results are highly encouraging, 99mTc-maraciclatide is still an investigational agent and not yet approved for clinical use. The next crucial step is to validate these findings in larger, more diverse patient populations. Serac Healthcare has announced that multi-centre, international Phase 3 studies are planned to begin later this year.
The agent has already been granted Fast Track Designation by the U.S. Food and Drug Administration (FDA), a process designed to expedite the review of drugs and diagnostics that address serious conditions and fill an unmet medical need. This designation could pave the way for a quicker path to regulatory approval if the Phase 3 trials are successful.
The potential impact extends far beyond just diagnosis. A reliable, non-invasive tool could revolutionize how endometriosis is managed. Clinicians could use it to monitor disease progression or recurrence without repeated surgeries. Perhaps most importantly, it could accelerate the development of new, desperately needed treatments. The global endometriosis market is projected to reach nearly $2 billion by 2032, a figure driven by a desperate need for innovation. By providing a clear, validated biomarker, the scan could serve as a reliable endpoint in clinical trials, making it easier and faster to test the efficacy of novel therapies and finally bring new options to patients who have waited far too long.
📝 This article is still being updated
Are you a relevant expert who could contribute your opinion or insights to this article? We'd love to hear from you. We will give you full credit for your contribution.
Contribute Your Expertise →