CDC Taps Cepheid to Build America's Next-Gen Pandemic Shield
- 5-year contract: The CDC-Cepheid partnership runs through February 2030, signaling long-term pandemic preparedness.
- Scalability: Cepheid must ramp up production from 10,000 tests/week to over 1 million during crises.
- 45-minute results: Cepheid’s GeneXpert system delivers rapid COVID-19 test results, a capability now expanded under the new initiative.
Experts would likely conclude that this CDC-Cepheid partnership represents a critical step toward building a more resilient and rapid-response diagnostic infrastructure for future pandemics, leveraging proven technology and pre-established frameworks to eliminate past bottlenecks.
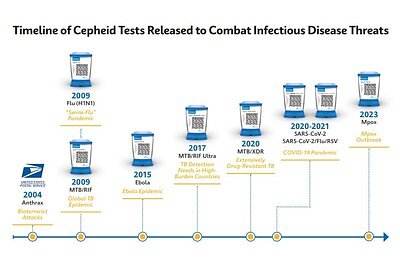
CDC Taps Cepheid to Build America's Next-Gen Pandemic Shield
SUNNYVALE, CA – February 18, 2026 – The U.S. Centers for Disease Control and Prevention (CDC) has selected diagnostics giant Cepheid as a key national partner in a sweeping new federal initiative designed to fortify the nation against future pandemics. The collaboration, part of a multi-company strategy, aims to dramatically slash the time it takes to develop and deploy accurate, high-volume tests in the face of a new pathogenic threat, addressing a critical vulnerability exposed during the early days of the COVID-19 pandemic.
Cepheid, a leader in molecular diagnostics, is one of four companies chosen for a long-term contract that formalizes a public-private alliance for rapid response. The award gives the company a direct line to the CDC's frontline data, including early access to outbreak samples and viral genomic sequences, creating a framework to turn a pathogen's genetic blueprint into a widespread diagnostic test in record time.
"For years, Cepheid has been at the forefront of outbreak response," said Vitor Rocha, President of Cepheid, in a statement accompanying the announcement. "We are honored to be selected by CDC for this IDIQ award for rapid, scalable diagnostics, and we are confident the collaboration can leverage our pioneering technology and global footprint to help public health systems respond swiftly and effectively to emerging challenges."
A New Blueprint for National Readiness
The partnership is structured under a multiple-award Indefinite Delivery-Indefinite Quantity (IDIQ) contract, a flexible tool that allows the government to secure services without pre-determining the exact scale or timing of future needs. According to federal procurement documents related to the initiative, the contract establishes a five-year performance period running through February 2030. This long-term framework signals a strategic shift from ad-hoc emergency contracts to building a permanent, sustainable infrastructure for diagnostic readiness.
The scope of the contract outlines a comprehensive and demanding set of expectations. Collaborators like Cepheid will be required to work with the CDC and the Food and Drug Administration (FDA) to create pre-approved development plans, ensuring that when a new threat emerges, the pathway to an Emergency Use Authorization (EUA) is already clearly defined. A crucial component of the agreement is the mandate for scalable manufacturing. Partners must demonstrate the ability to ramp up production from an initial 10,000 tests per week to over one million, ensuring that a validated test can be distributed nationwide to meet crisis-level demand.
This structured approach is a direct lesson learned from past outbreaks, where logistical hurdles, regulatory bottlenecks, and a lack of pre-established relationships between government and industry led to costly delays. By creating this network of trusted partners in peacetime, the CDC aims to build a more resilient and coordinated national response capability.
From Genetic Code to Clinic in Record Time
At the heart of the initiative is the goal of radical acceleration. Cepheid’s selection hinges on its proven ability to rapidly translate viral data into a functional diagnostic tool, a capability demonstrated during the COVID-19 crisis. In March 2020, the company secured the first FDA Emergency Use Authorization for a point-of-care PCR test, delivering results in approximately 45 minutes on its widely deployed GeneXpert systems. This was achieved within weeks of the SARS-CoV-2 sequence being published.
The new CDC collaboration is designed to replicate and improve upon that success. By providing early access to critical reference materials and pathogen sequences, the CDC will enable Cepheid to begin the work of designing and validating primer and probe sequences—the key components of a PCR test—almost immediately upon threat identification. This eliminates a significant bottleneck that previously hindered private labs.
Cepheid’s GeneXpert platform is a key element of this strategy. The system consists of automated, self-contained instruments that are already installed in nearly 5,000 U.S. locations, from major urban hospitals to remote clinics. This existing footprint allows for decentralized testing, meaning a new diagnostic can be rapidly deployed across the country without requiring samples to be sent to a limited number of central labs. The platform’s versatility is also a major asset, with a broad existing portfolio of tests for infectious diseases like tuberculosis, influenza, and HIV, showcasing its reliability and adaptability.
"Cepheid has proven we can reliably deliver quality molecular diagnostics when they're needed most," noted Larry Kelmar, the company's Vice President of Government Programs and Pharma Collaborations. "Working closely with partners like the CDC strengthens our ability to rapidly respond to new outbreaks and safeguard communities with advanced, high-impact testing solutions."
The Business of Preparedness
This selection solidifies Cepheid's position as a cornerstone of U.S. public health infrastructure and provides significant long-term business visibility. The IDIQ contract ensures a sustained partnership and a potential stream of revenue tied to national security and health priorities. For investors and the market, it signals federal confidence in the company's technology and its ability to deliver under pressure, reinforcing its competitive advantage in the molecular diagnostics space.
However, the CDC’s strategy is not to rely on a single champion. By selecting four distinct collaborators, the agency is building redundancy and fostering a competitive ecosystem designed to spur innovation. While the other three partners under this specific rapid diagnostic development contract have not yet been publicly detailed, the CDC has announced related collaborations with major laboratory players like Labcorp and Quest Diagnostics for expanded testing capacity and data sharing. This multi-pronged approach indicates a broader strategy to create a distributed and resilient network of diagnostic capabilities across the private sector.
This public investment is expected to shape the entire diagnostic industry, pushing competitors to bolster their own rapid response platforms and seek similar government partnerships. The era of pandemic preparedness is creating a new market where speed, scalability, and reliability are the most valuable currencies. As Kelmar added, "CDC's selection reflects not only our technical expertise but also the critical need for sustained collaboration to ensure the country is ready for whatever comes next."
