BrightGene's New Drug Redefines Obesity Treatment Beyond Weight Loss
- Weight Loss: 19.3% average reduction in body weight
- Blood Pressure Improvement: 22.9 mmHg systolic and 12.9 mmHg diastolic reduction in hypertensive patients
- Bone Health Boost: 3.9% increase in hip bone mineral density
Experts view BGM0504 as a groundbreaking obesity treatment that goes beyond weight loss, offering comprehensive cardiometabolic benefits and superior tolerability, potentially redefining the management of obesity-related diseases.
BrightGene's New Obesity Drug Redefines Success Beyond Weight Loss
SUZHOU, China – May 06, 2026 – Chinese biotech firm BrightGene Bio-Medical has unveiled striking Phase III clinical trial results for its weight-loss drug, BGM0504, demonstrating not only potent efficacy that rivals market leaders but also a sweeping range of health benefits that could redefine the treatment of obesity and related metabolic diseases.
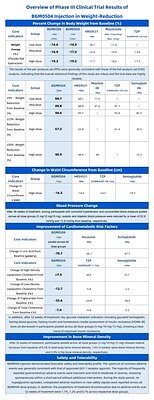
The data, from a 52-week study involving 652 participants, showed that patients on the highest dose of the GLP-1/GIP dual agonist achieved an average body weight reduction of 19.3%. But in a market where powerful weight loss is becoming the norm, it is the drug's comprehensive impact on overall health and its remarkable tolerability that sets it apart.
A Paradigm Shift Beyond the Scale
For years, the race in obesity therapeutics has been a contest of percentages—a singular focus on who can deliver the highest number in weight reduction. BrightGene's results suggest a strategic shift in that paradigm, moving the goalposts from simply shedding pounds to achieving comprehensive cardiometabolic regulation.
Beyond the impressive 19.3% weight loss and a 16.5 cm reduction in waist circumference, the trial data revealed significant improvements across a spectrum of critical health markers often associated with obesity:
- Blood Pressure: In a subset of patients with comorbid hypertension, BGM0504 delivered a stunning mean reduction in systolic and diastolic blood pressure of 22.9 mmHg and 12.9 mmHg, respectively. This effect is comparable to results often requiring dual or even triple combination therapy with standard antihypertensive drugs. An impressive 92.9% of these patients reached their target blood pressure goals.
- Uric Acid: Participants saw a mean reduction in uric acid of 70.7 μmol/L. This is a clinically significant drop, placing it within the therapeutic range of dedicated gout medications like febuxostat and potentially reducing the risk of painful gout attacks.
- Blood Lipids: The drug prompted a 33.6% reduction in triglycerides and a 12.7% drop in LDL ("bad") cholesterol, outperforming some competitors on these metrics. This was coupled with a 7.3% increase in HDL ("good") cholesterol, indicating a holistic improvement of the lipid profile.
These multi-faceted benefits suggest BGM0504 is not just a weight management tool but a powerful agent for remodeling a patient's entire metabolic state, tackling the interconnected web of conditions known as cardiometabolic disease.
"We believe that an ideal weight management therapy should not be narrowly confined to the singular metric of weight reduction, but rather, should focus on the comprehensive regulation of cardiometabolic health," stated Dr. Yuan Jiandong, Chairman of BrightGene Bio-Medical, in the company's announcement.
Challenging the Titans on Efficacy and Tolerability
BGM0504 is entering a fiercely competitive arena dominated by pharmaceutical giants like Eli Lilly and Novo Nordisk. While its 19.3% weight reduction places it squarely in the top tier, competitive with Eli Lilly's tirzepatide (Zepbound) and surpassing Novo Nordisk's semaglutide (Wegovy), its true competitive edge may lie in who can take the drug and for how long.
A key differentiator is the drug's exceptional tolerability. The discontinuation rate due to adverse events in the high-dose group was a mere 0.7%. This figure is dramatically lower than the 6-7% discontinuation rates reported in trials for some competing products. This suggests that patients find BGM0504 significantly easier to tolerate, a critical factor for long-term adherence to a chronic medication. The common gastrointestinal side effects were reported as mostly mild and transient.
Furthermore, the drug demonstrated strong efficacy even at its lowest dose, with the 5mg group achieving a 14.3% mean weight reduction. This "low-dose efficacy with dose-dependent enhancement" allows physicians to start patients on an effective dose that builds confidence while minimizing initial side effects, a significant advantage in real-world clinical practice.
The results also highlight the drug's ability to induce "deep weight loss." Nearly half (48.9%) of the participants on the highest dose lost over 20% of their body weight, an outcome that begins to approach the efficacy of bariatric surgery. Professor Ji Linong, the trial's principal investigator, expressed his satisfaction with these outcomes, underscoring their clinical significance.
An Unexpected Boon for Bone Health
Perhaps the most surprising and potentially significant finding from the trial was BGM0504's effect on bone mineral density (BMD). Significant and rapid weight loss, whether through diet or medication, is often associated with a reduction in bone density, raising long-term concerns about increased fracture risk.
In a remarkable departure from this trend, patients treated with BGM0504 showed an increase in bone density across the board. After 52 weeks, participants saw a 1.4% increase in lumbar spine BMD and an even more impressive 3.9% increase in hip BMD.
This counterintuitive finding suggests that the drug's metabolic effects may extend to actively preserving or even strengthening the skeleton during weight loss. If this benefit is confirmed in further long-term studies, it would represent a major safety advantage over other therapies and address a key concern for physicians and patients considering long-term use of these powerful medications.
The Path to Market and Patient Experience
With these robust Phase III results in hand, BrightGene plans to submit a New Drug Application to China's National Medical Products Administration (NMPA) for chronic weight management. While the initial focus is on the vast Chinese market, the company's presentations at international conferences suggest global ambitions.
For patients, the promise of BGM0504 extends beyond the number on a scale. The data paints a picture of a treatment journey that is not only highly effective but also more tolerable and holistically beneficial. The potential to lose significant weight while simultaneously improving blood pressure, cholesterol, and uric acid levels—all without compromising bone health and with a lower likelihood of disruptive side effects—could fundamentally change the experience of managing obesity.
As the medical community increasingly views obesity as a complex, chronic disease at the root of numerous other health problems, therapies like BGM0504 that can tackle multiple risk factors at once are poised to become a cornerstone of modern medicine. The drug’s comprehensive profile offers a promising new tool for the multidimensional clinical management of cardiometabolic diseases, shifting the focus from simply losing weight to gaining health.
📝 This article is still being updated
Are you a relevant expert who could contribute your opinion or insights to this article? We'd love to hear from you. We will give you full credit for your contribution.
Contribute Your Expertise →