BioCardia Advances Key Device for Precision Heart Treatment
- FDA accepted BioCardia's pre-submission package for the Helix Transendocardial Delivery Catheter in March 2026
- BioCardia's market capitalization: approximately $13.5 million
- CardiAMP Cell Therapy received FDA Breakthrough Therapy Designation, enabled by Helix
Experts view the FDA's acceptance of BioCardia's pre-submission package as a significant validation of the Helix catheter's potential to advance precision heart therapy delivery, though they caution that the company's financial stability and regulatory success remain critical factors in its long-term viability.
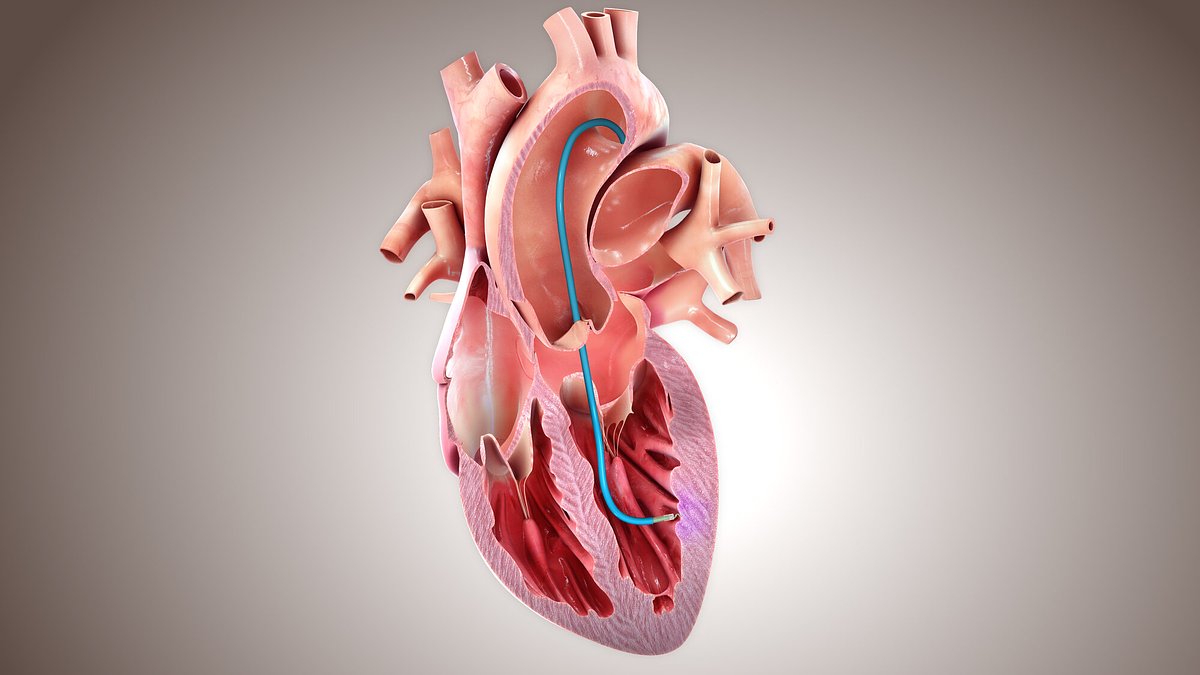
BioCardia Advances Novel Device for Precision Heart Therapy Delivery
SUNNYVALE, CA – March 17, 2026 – BioCardia, Inc., a developer of advanced cardiovascular therapeutics, has taken a significant step forward in its mission to reshape heart disease treatment. The company announced today that the U.S. Food and Drug Administration (FDA) has accepted its pre-submission package for the Helix Transendocardial Delivery Catheter, a device poised to solve one of the most persistent challenges in cardiology: delivering therapies directly and precisely to a beating heart.
This regulatory milestone sets the stage for a substantive review meeting with the FDA in the second quarter of 2026. While the announcement represents a crucial validation for the California-based biotech, it also magnifies the high-stakes journey of a company navigating complex clinical trials and tight financial pressures in its quest to bring next-generation cardiac treatments to market.
A New Tool for a Pressing Need
For decades, physicians have struggled with the limitations of delivering treatments for heart disease. Systemic drug administration often results in therapies being diluted throughout the body, with only a fraction reaching the intended target in the heart muscle, or myocardium. This inefficiency is a major roadblock, especially for the burgeoning field of cardiac cell, gene, and protein therapies, which require high concentrations at the site of injury to be effective.
The Helix catheter is BioCardia's answer to this problem. It is a minimally invasive system designed to be guided through the body’s vasculature into the chambers of the heart. Its defining feature is a specialized, small helical needle at its tip. This unique design allows a physician to gently engage the catheter with the inner wall of the heart muscle. This provides critical stability within the dynamic, constantly moving environment of a beating heart, enabling the precise and controlled injection of therapeutic agents into targeted areas of the myocardium.
According to the company, this method ensures superior retention of the therapeutic agent compared to other delivery modalities. It also allows access to regions of the heart that are difficult or impossible to reach with other methods, potentially opening up new treatment possibilities for patients with extensive heart damage.
“FDA marketing clearance of the Helix would be meaningful for our business as the entire field of cardiac cell, gene, and protein therapeutics needs an FDA approved minimally invasive percutaneous intramyocardial delivery system,” said Peter Altman, PhD, BioCardia's Chief Executive Officer, in a statement.
Navigating a Complex Regulatory Pathway
The FDA's acceptance of the pre-submission package is the first formal step on a long and intricate regulatory road. The upcoming meeting will involve both the FDA's Center for Devices and Radiological Health (CDRH), which oversees medical devices, and the Center for Biological Evaluation and Research (CBER), which regulates cell therapies and other biologics. This dual-center consultation is critical, as the Helix catheter is designed as a delivery vehicle for biological products, making it a key component of a combination therapy.
The path to market clearance will depend on the device's final classification by the FDA. If deemed a lower-risk Class II device with a comparable predecessor on the market, it could pursue a 510(k) clearance, a process that can take roughly six months. However, given its novelty and role in delivering breakthrough therapies, it may be classified as a higher-risk Class III device, requiring a more rigorous and lengthy Premarket Approval (PMA) process that often takes a year or more. A third possibility is the De Novo pathway for novel devices without a predicate, which also involves a thorough review.
The involvement of both CDRH and CBER underscores the device's integral role in the therapies it's designed to deliver. A positive review could not only clear the path for the Helix catheter as a standalone device but also streamline the review process for the therapies that depend on it.
The High-Stakes Gamble for a Small Biotech
While the regulatory progress is a significant scientific and clinical validation, it occurs against a challenging financial backdrop for BioCardia. With a market capitalization of approximately $13.5 million, the company is a small player in the fiercely competitive biotech arena. Its stock has seen a significant decline over the past year, and financial filings indicate a cash runway extending only into the current quarter, Q2 2026.
This financial pressure makes every milestone critical. The announcement has been met with cautious optimism from market watchers. While some analysts see the company's technology as undervalued, others point to the inherent funding risks associated with a clinical-stage company that has yet to generate meaningful revenue. Successfully bringing a device like Helix to market could be transformative, creating a new revenue stream and establishing BioCardia as a key partner for other companies developing cardiac biotherapeutics.
Failure to secure clearance or the need for additional, costly trials could, however, place significant strain on its limited resources. The company's future hinges on its ability to successfully execute on its clinical and regulatory strategy while securing the necessary funding to see its projects through to completion.
An Enabling Platform for Next-Generation Heart Therapies
The true significance of the Helix catheter is inextricably linked to BioCardia's lead therapeutic candidate, the CardiAMP Cell Therapy. This autologous therapy, which uses a patient's own bone marrow cells to treat ischemic heart failure, relies entirely on the Helix system for delivery. The FDA has already acknowledged this synergy by granting a Breakthrough Therapy Designation for the CardiAMP treatment, a designation that was explicitly "enabled by Helix."
The CardiAMP program has shown both promise and challenge. A Phase 3 trial, CardiAMP-HF, demonstrated positive signals in two-year outcomes, including increased survival and reduced major adverse events for certain patient subgroups. However, the trial was paused in 2023 after an interim analysis projected it was unlikely to meet its primary one-year efficacy endpoint, a significant setback. BioCardia has since been in discussions with the FDA about a potential protocol redesign.
This is where the approval of the Helix catheter becomes a pivotal strategic goal. Securing FDA clearance for the delivery system would de-risk a major component of the overall CardiAMP therapy platform. It would provide a validated, approved tool that could then be used in future CardiAMP trials, potentially simplifying the therapy's own path to final approval. The company is actively pursuing this, with a separate FDA submission on the CardiAMP approval pathway also expected this quarter, alongside upcoming clinical data presentations in Europe. The fate of the device and the therapy it enables are now more closely intertwined than ever as BioCardia pushes forward on multiple fronts.
